Trust Agency
Trust Agency
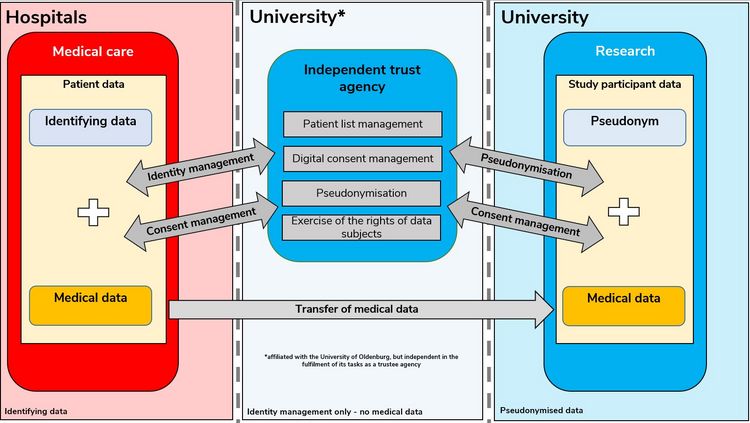
Establishment of an independent trust agency
A central independent (data) trust agency is currently being established at the University of Oldenburg. The trust agency is intended to make a further service contribution to supporting data protection-compliant personal research, especially in the area of the school of Medicine, at the University of Oldenburg.
For this purpose, the trustee agency shall, among other things, provide software solutions that enable the systematic administration of personal identifying data and consent declarations. In particular, the independent trust agency shall perform the following specific tasks:
- Development and operation of a system for digital consent management.
- Development and operation of a system for maintaining digital patient lists
- Pseudonymisation service
- Ensuring inter-institutional data linkability
- Contact point for the exercise of data subjects' rights
- Duplicate entry resolution
Uniform interfaces for local aspects of identity and consent management will be provided for researchers and hospitals. Local tasks include, for example, registration of study participants or obtaining informed consent from study participants by staff at the respective institutions. These interfaces should also enable standardised and systematised information on whether and which consents are stored for a study participant and in which context the data may be accessed for research purposes. Consents may be broad informed consent or specific informed consent.
The independent trust agency will only grant access to systems and data based on a system of roles and rights. Hospitals, university institutions, project partners and staff will only have access to data to which they are authorised. The systems are operated under suitable security precautions in the data centre of the University of Oldenburg. Medical data is not processed in the trust office and the employees of the trust agency do not have access to research data.
The figure Identity management infrastructure with the independent trust agency at the Oldenburg site shows an example and simplifies the planned interaction between hospitals, the University of Oldenburg and the independent trust agency in the transfer of medical data into the research context. The technical implementation of data flows and processes may vary depending on the project or study.