We think of the cornea as a windscreen for the eye. But this transparent layer of tissue is far more complex than a simple protective covering. Stefan Schrader and Sonja Mertsch are pursuing new approaches to treating diseases of the ocular surface.
Just a few years ago there would have been no way to help the eighty-year-old patient. The elderly woman had an ulcer on the cornea of her eye, probably caused by diabetes. Prof. Dr. Dr. Stefan Schrader and his colleagues at the University Hospital Düsseldorf, where he was still working at the time, had already tried everything: for several months the patient had taken special eye drops and the doctors had also transplanted a so-called amniotic membrane onto areas that were not healing properly several times. This membrane, which is extracted from human placenta, functions like a dres-sing and promotes wound healing.
But the treatment wasn’t working. The patient was facing the prospect of losing her eye. “All the conventional treatments had failed,” explains Schrader, who since October 2018 has been Professor of Ophthalmology at the University of Oldenburg and Director of the University Eye Clinic at the Pius-Hospital Oldenburg. The lack of alternatives forced the specialists to make an unusual decision in consultation with the elderly patient: instead of performing yet another transplantation – the standard treatment in cases of acute corneal disease – the team used an artificial membrane based on collagens. These structural proteins form fibres and are an integral part of the tissue in the skin, bones and tendons. “We administered the colla-gen construction as a combination of basal-membrane substitute and dres-sing. This new procedure had a most impressive effect,” Schrader says. The wounds in the cornea healed and after six months there were no signs of inflammation or irritation in the artificial material. The patient not only kept her eye but was also able to see more clearly again.
The cornea might seem unremarkable at first glance, but it plays an essential role. The small, transparent disc is extremely thin and about the size of a ten-cent coin. It forms the centre of the eye surface and covers the iris and pupil. The cornea borders with the Ophthalmologysclera, which encloses the rest of the eye. The tissue consists of six micros-copically thin layers, the thickest of which is the stroma, which contains collagen. “The exterior layer of the cor-nea, which is moistened by lacrimal fluid, protects the interior of the eye like a windscreen,” Schrader says – against germs, foreign bodies and UV light for example. The cornea also provides almost three-quarters of the eye’s refractive power. Together with the lens, it helps the eye to optimally focus incoming light so that a sharp image forms on the retina.
When donor corneas don't help
Diseases of the cornea can have drastic consequences. “If scarring occurs as a result of bacterial or viral infections, injuries or various diseases, the cornea first of all loses transparency, and secondly, its refractive power is altered. Both these factors impair vision and can lead to total loss of eyesight. Transplantation of donor tissue can prevent complete loss of vision in most patients suffering from severe corneal diseases. Schrader performs such operations regularly at the Pius-Hospital. In some cases, doctors replace the entire cornea, in others they perform microsurgery to replace only the damaged area.
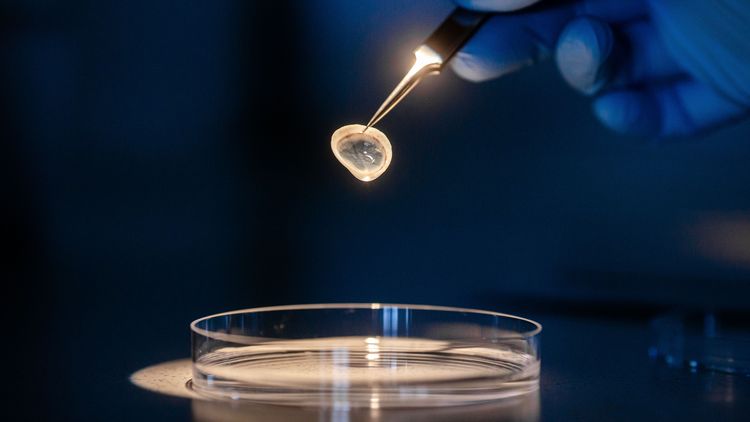
But this doesn’t work for all patients. On the one hand, severe disorders of the ocular surface – for example dry eye or damage to the corneal nerves – can hinder transplantation or even render it impossible. On the other hand, there might be a shortage of donor corneas, explains Dr. Sonja Mertsch, a neurobiologist and head of the research laboratory for experimental ophthalmology on Wechloy Campus. She and Schrader are therefore investigating new options for reconstructing the ocular surface, for example the collagen gel used to treat the eighty-year-old patient, which was created at Mertsch and Schrader’s previous workplace, the University Hospital Düsseldorf. “The material is highly promising for research,” emphasises Mertsch, explaining that like other types of connective tissue, the cornea consists primarily of collagen. But commercially available gels have a disadvantage in clinical practice: their viscous consistency means they can’t be surgically attached to the eye. So the researchers decided to get creative and compressed the gel to remove its liquid content.
The experiment paid off: “It is indeed possible to sew this new biomate-rial and use it as a transplant,” Mertsch reports. Further tests followed in the lab – for example to find out more about the gel’s elastic properties and biocompatibility. “We tested whether corneal cells degrade the gel, whether it is harmful for the cells, and whether they can proliferate on it,“ the neurobiologist explains. The researchers found no evidence of negative effects in their experiments. Since the collagen used to produce the gel had already been approved for human use in orthopaedics, there was nothing to prevent it from being used in ophthalmology. After the first successful treatment, Schrader now plans to carry out a clinical study in collaboration with the University Hospital Düsseldorf.
Replacement for damaged corneas
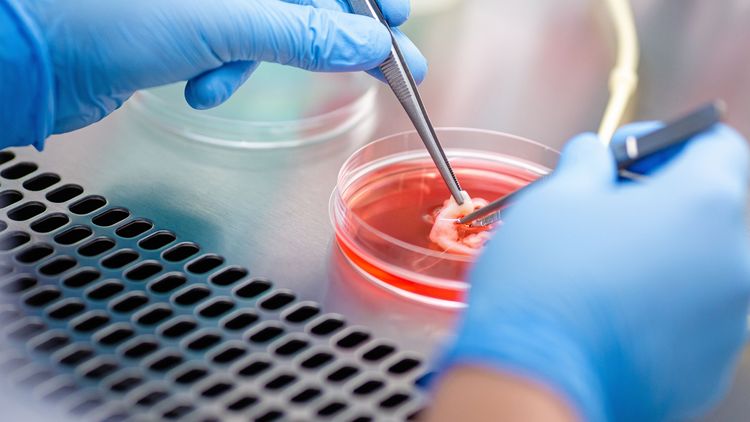
With the new laboratory for experimental ophthalmology on Wechloy Campus, Mertsch has already laid the foundations for this. Using state-of-the-art technology, the scientists are currently investigating various ways to improve ocular surface reconstruction. “We have everything we need here to produce new biomaterials and analyse their function and structure. We can also test the effects of drugs – all the way through to DNA and protein analysis,” she says. For example, Mertsch and her team are using a certain type of corneal cells, so-called fibroblasts, to produce repla-cement material for damaged corneas. Under certain conditions fibroblasts occur in the cornea, where they produce the collagen. For their experiments the eye researchers are using cells from donated corneas that are not suitable for transplantation. Schrader and Mertsch have already developed a procedure that makes these cells grow into thin layers or “sheets”. The advantage of these so-called “cell sheets” is that they resemble natural corneal tissue much more closely than the biomate-rials currently in use.
Unusually high numbers of nerve cells
After just three weeks the cell sheets are around forty micrometres thick, that is four hundredths of a millimetre, and after one year they grow to a thickness of approximately 150 micrometres. In the future, patients could potentially have their own fibroblasts removed to make tissue for corneal reconstruction. “This would minimise the risk of rejection,” Schrader says. Tests have produced positive results so far: the new material is extremely transparent, strong enough to with-stand an operation, and corneal cells have no problem growing on it. The team’s current objective is to use growth factors – special molecules that stimulate cellular growth and proliferation – to reduce the time the tissue needs to reach the desired level of thickness to about three months.
Whether or not a transplant using artificial or donor tissue succeeds also depends on the nerve cells in the eye: “The cornea has an unusually high number of nerve cells,” Schrader explains. We only become aware of the functionality of these neurons when, say, we get a fly in our eye when cycling. We immediately feel a burning pain. The eyelid closes and lacrimal fluid shoots into the eye. “The nerve cells trigger a blinking reflex to keep the ocular surface intact. They also control secretion of the lacrimal glands and other glands which moisten the eye surface,” the researcher explains.
After a corneal transplant, the nerve cells grow into the new tissue again, but even twenty years later their numbers still won’t reach the original levels in many patients, Mertsch says. The problem here is the close interaction between nerve cells and corneal cells. If the nerve cells are not working properly, the cells on the surface of the cornea will atrophy. In the worst case they will die, and this can result in further damage to the eye surface. Herpes infections, laser operations on the cornea, brain tumours and diseases like diabetes or multiple sclerosis can also negatively impact nerve function to such a degree that the outer layer of the cornea is damaged. “This is why we are trying to find drugs that stimulate nerve growth in the cornea,” Mertsch explains.
To do this the researchers need a proper model of the corneal tissue. After years of research the neurobio-logist has succeeded in reproducing the tissue in the lab – using as a basis the collagen gel that gave such good results as a wound bandage. To this base all the main cell types from the various layers of the cornea are added. “We have combined all the individual components into a unified structure and this has given us a model of the cornea as a whole,” Mertsch says. The trickiest part was to get the nerve cells to grow in the lab. Using state-of-the-art molecular biological methods, the researchers are now studying which drugs influence nerve growth and which metabolic processes play a key role. They have already tracked down a number of interesting substances, the neurobiologist says.
Regenerating damaged nerves
To test the effectiveness of these substances the team is conducting various experiments, some of them on mice. Here the researchers operate the mice cornea just as they do for a human corneal transplant, and follow up with a four-week course of eye drops. Through these tests they have identified an active substance that is particularly effective in stimulating regeneration in damaged nerves. “This substance has already been approved as a drug for another eye disease. This is a lucky break for us because it means we will soon be able to start performing patient studies,” Mertsch adds.
In the search for new treatment methods the scientists contemplate the entire ocular surface as a functional unit – including the eyelids, eye-lashes and the lacrimal gland. “When one subsystem fails the entire unit is affected,” says Schrader. The lacrimal gland plays an important role here: if it doesn’t release enough fluid the eye surface dries out, sustains damage and can become inflamed. Ulcers form and vision is impaired. “Dry eye” is a very common condition and is cau-sed by aging of tissues, autoimmu-ne diseases and external factors such as long hours in front of the computer screen. The symptoms of dry eye can be treated using tear substitutes, so-called artificial tears. “But we are still unable to regenerate the damaged lacrimal gland. This means we lack curative treatment methods, or in other words the possibility to heal the disease,” Schrader emphasizes. But here, too, the Oldenburg ophthalmologists have come up with some promising approaches.
Stem cells for the lacrimal gland
The researchers are using mesenchymal stem cells, for instance. These cells are found in the bone marrow or fatty tissue of human adults and, like em-bryonic stem cells, they can divide and transform into other cell types. Jana Dietrich, a doctoral student in the lab for experimental ophthalmology, has already shown in her doctoral thesis that these stem cells have a therapeutic effect on the damaged lacrimal gland in mice. To verify these results the resear-chers now plan to work with human mesenchymal stem cells. They are able to get these cells from a stem cell bank.
Alongside the work with stem cells, the researchers are trying to replace the lacrimal gland with artificial tissue, as they did with the cornea. As a scaffold for their experiments they are using sections of pig intestines from which all animal cells have been removed. The idea is to grow human cells on it in a special bioreactor. The researchers have already succeeded in delivering a constant supply of nutrients to the constructs in the bioreactor under special cultivation conditions, similar to what takes places in the body via the blood vessels. This cultivation ensures that the lacrimal gland cells grow on the construct in several layers and are functionally active. “That was a major breakthrough,” Mertsch says. “The new tissue even produced a form of lacrimal fluid.” One challenge the researchers have yet to overcome, however, is the different conditions for growth which the many different types of cells of the lacrimal gland require.
But these are not the only reasons why an artificial organ is still a distant goal: porcine intestinal tissue is simply too large to provide a good basis for a human lacrimal gland. Moreover, an artificial gland would need to be connected to nerves that control tissue activity. “This is definitely a long-term project,” Mertsch emphasizes. But the researchers are optimistic. In their new laboratory she and Schrader have everything they need to work on their many research questions. And they are clearly focused on their objective: to help and maintain vision in patients for whom conventional corneal transplants are out of the question.