Kontakt
Anschrift
Neurobiologie des Sehens
Department of Neuroscience
Carl von Ossietzky University Oldenburg
Carl-von-Ossietzky-Straße 9-11
26129 Oldenburg
Contact
Research

Research
How are visual signals transmitted from the retina to the brain?
The goal of our research is to understand visual signal processing in the retina. Understanding how populations of neurons encode information is a central problem in neuroscience. The retina is an ideal system to study this question because its natural inputs can rigorously be determined and experimentally controlled, and because it is now possible to access and record the activity of a large fraction of its output neurons simultaneously. Furthermore, the anatomy and functional organization of the retina are remarkably well known, which strongly constrains models of information processing.
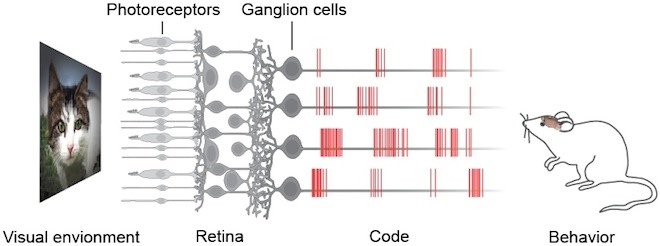
The visual stimulus is transduced into electrical signals by the photoreceptors of the retina, processed by many types of interneurons, and conveyed to the brain by the ganglion cells along their axons that form the optic nerve. All visual information is encoded in temporal patterns of electrical impulses of the ganglion cells. We know from functional and morphological studies that about 30 types of ganglion cells exist. The distinct types show different light response properties, sizes, and branching patterns. Each type carries a specialized and distinct representation of the visual environment to different targets in the brain. We do not know the detailed response properties of many types, especially in natural viewing conditions, nor do we know how the diverse information sent by the different cell types collectively determines visual perception and behavior.
Electrophysiology
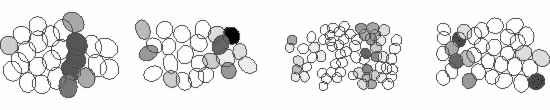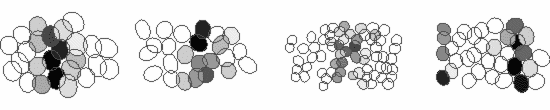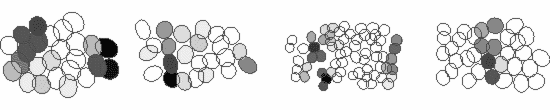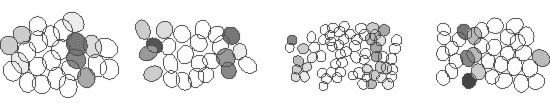
We study these questions using large-scale multi-electrode arrays, which are able to record the responses of several hundred neurons of the various cell-types simultaneously. For example, shown below are the responses of four ganglion cell types to a stimulus grating moving across the retina. Each ellipse represents the receptive field of a cell and the activity of the cell is shown by the darker colors. The receptive fields of ganglion cells of the same type tile the retina with a uniform overlap, thereby ensuring that every point of the visual space is covered by at least one cell of each type. Thus, each ganglion cell type conveys a full visual image to a distinct set of targets in the brain, suggesting that they serve different visual functions.
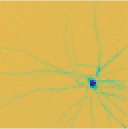
The high number and density of electrodes of the array make it possible to construct a footprint of the recorded cells. This footprint is the average voltage the electrodes record when the cell fires an impulse. Shown below, the footprints of a ganglion cell (left) and a polyaxonal amacrine cell (right) on the array. In the case of the ganglion cell, the impulse migrates along the axon toward the blind spot. In the case of the amacrine cell, the impulses travel across the retina along multiple axons. Amacrine cells play an important role in the lateral inhibition of ganglion cells and thereby in the formation of the response properties of the various types.
 |  |  |  |
Anatomy
We perform anatomical experiments to further investigate the remarkable diversity of ganglion cells and their presynaptic partners by combining immunohistochemistry, single-cell dye injections, and confocal microscopy. Cell markers for distinct types retinal ganglion, amacrine, or bipolar cells allow the analysis of their distribution within the retina and putative interactions between the types. These experiments help to elucidate type-specific features of the cells and how the type-dependent synaptic circuitries shape the unique light response properties of individual ganglion cell types. The tissue used for multi-electrode recordings can subsequently be processed for anatomical studies and the recorded cells can be mapped to their immunological identity as determined by antibody stainings.
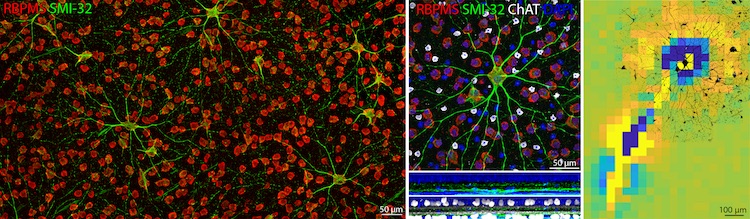
The left panel of the figure above shows an antibody labeling of all ganglion cell bodies (red) and specific ganglion cell types (green) in a region of the guinea pig retina. An ON sustained alpha ganglion cell is shown in the middle panel (green, top). The bottom panels depict the stratification level of the cell’s dendrites in the inner plexiform layer relative to all cell nuclei (blue) and starburst amacrine cells (white). In the right panel, the same alpha ganglion cell (black) was matched onto its electrical footprint from the preceding multi-electrode recording.

The left panel of the figure above shows a confocal image of a labeled ganglion cell (green) in the mouse retina which is positive for a marker of alpha ganglion cells (red). Each dot in the middle panel represents the location of a ganglion cell body in a complete mouse retina. The right panel shows a transformation of the same data into a sphere. Darker colors indicate higher local densities of ganglion cells.

