Contact
Head
Prof. Dr. Niklas Nilius
Secretary
Meike Hurling
Kontakt
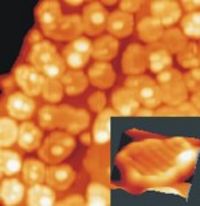
Metal Nanoparticles
Growth and Characterization of Metal Nanoparticles
- Size and shape of metal particles governs their electronic and optical properties as well as their chemical (catalytic) response
- Particle nucleation on oxides preferentially occurs on defects and domain boundaries
- Oxide doping enables control of the particle shapes
Metal Nanoparticles on MgO(001)
- Particle geometry determined by interfacial adhesion, ionization potential of ad-metal and lattice match with the oxide support
- Unusual crystal structures of nanoparticles to maximize metal-oxygen bonds across the interface
Gold Particles on Mo-doped CaO(001)
- 3D growth of gold on pure CaO due to small metal-oxide adhesion
- Charge transfer from Mo-dopants into gold increases adhesion and triggers formation of 2D particles