Hydrogen detection film
Welcome to the hydrogen detection film overview page
The materials provided here were created in co-operation with the working group of Prof. Stefan Bernhard and his PhD student Savannah Talledo. They are part of Malte Petersen 's dissertation project in our research area of specialised didactic transfer research.
On this page you will find background information, materials for use in the classroom and further publications. If you are interested in receiving a sample for your classroom free of charge, please contact us using the form below or contact us directly by email.
The site is constantly under construction and will be expanded step by step to include further possible uses and instructions for the classroom. You are welcome to contact Malte Petersen with further ideas for use in the classroom.
Download link for the teaching materials:
How the detection film works
The hydrogen detection film turns dark blue within a very short time on contact with hydrogen. Storage in the ambient air causes a re-reaction with the atmospheric oxygen, the film regenerates and can be used again.

Use of the verification film in the classroom
The hydrogen detection foil offers the possibility of extending various experiments at school. By using PDMS as a carrier material, the detection film can be used both in the gas atmosphere and directly in the reaction solution. When added to the reaction solution, it can detect the hydrogen formed during the reaction of a base metal with an acid or an (earth) alkali metal with water within less than a minute (see illustration). Alternatively, a detection foil can be attached to the bottom of a test tube with superglue and used in addition to the oxyhydrogen test. The blue colouring of the detection foil not only detects the hydrogen, but also gives an indication of when the oxyhydrogen gas sample is positive.

Introduction of the hydrogen detection film (the detection)
To introduce the hydrogen detection foil, it can be tested in different gas atmospheres before use in an experiment. To do this, different films are placed in suitable glass containers and these are filled with different gases one after the other. The detection foil only changes colour when hydrogen gas is used.
The detection foil in addition to the oxyhydrogen sample
The hydrogen detection foil can also be used in addition to the oxyhydrogen sample. In preparation for this, the foil is attached to the bottom of the test tube with superglue (see image) and this is then used to collect the hydrogen. The foil offers two advantages. On the one hand, it provides additional evidence of the hydrogen through the colour indication and, on the other hand, it is an indicator of when enough hydrogen has been collected for a positive oxyhydrogen sample.

Demonstrating the volatility of hydrogen
The volatility of hydrogen is considered in the classroom, for example, when looking at a future hydrogen economy and must be included in this for a balanced assessment. The detection film can be used in a simple experiment to impressively demonstrate the challenges of storage and transport.
To do this, a balloon is filled with hydrogen and detection films are attached at various points with transparent adhesive tape. After just a few minutes, the detection foil begins to change colour and indicates the hydrogen flowing out of the balloon (see picture).

Mechanism of the hydrogen detection film
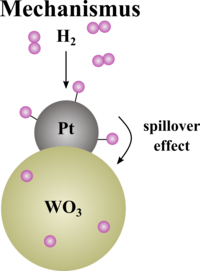
The hydrogen detection foil consists of polydimethylsiloxane (PDMS) and contains platinum-loaded tungsten(VI) oxide as the active material for hydrogen detection. The system is based on the hydrogen spillover mechanism, in which hydrogen molecules dissociate on the surface of the platinum catalyst and are then transferred into the metal oxide structure. The hydrogen atoms are embedded (intercalated) in the tungsten(VI) oxide and form the deep blue tungsten bronze. This visible colour shift is related to the formation of W5+ ions, which leads to a mixture of +V and +VI oxidation states and enables intervalley charge transfer transitions (IVCT) in the visible light range. Reduced evaluation tasks for the school can be found in the material folder provided.
A more detailed description can be found in the following publications:
- Petersen, M., Talledo, S., Bürger H., Bernhard, S., Wilke, T. (2026). Sample without bang: A reusable hydrogen detection foil for school lessons. CHEMKON 33(1), 11-16. doi: https://doi.org/10.1002/ckon.202500029
Reusability of the detection film
The hydrogen detection film can be regenerated as often as required and then reused. In our tests, we have reused individual films over 50 times and have not been able to determine any restriction in functionality. However, it is theoretically possible for defects to form over time. These should not restrict the functionality of the detection film.
Resistance of the detection film
Deactivation of the detection foil:
In the first step, the hydrogen detection foil is based on the reaction of hydrogen with the catalytically added platinum. This can be poisoned by known catalyst poisons such as carbon monoxide or ammonia, which severely restricts or prevents the functionality of the detection foil. If the platinum is poisoned, the film can be stored in an oxygen atmosphere and thus regenerated.
Reaction conditions:
The detection foil consists of a detection material that is integrated into PDMS(polydimethylsiloxane). PDMS is a silicon-based polymer and is considered chemically inert. The film can therefore be used in most systems at school. Under very extreme conditions such as the use of a strong base + heating, a high concentration of hydrogen peroxide + UV light or organic reagents such as toluene, hexane or chloroform, damage to the detection film can occur over time.
Colourants:
When using the detection foil in reaction systems with dyes, these should be checked for their influence beforehand. Dyes such as Nile red, methylene blue or methly red, for example, can colour the detection film and thus mask the actual colouring by the detection.
Density of the detection film
The hydrogen detection film consists of polydimethylsiloxane (PDMS), which generally has a density of 0.965 kg*m-3. By integrating the detection material, the density is further increased and approaches that of water. By shaking the liquid together with the detection film, it is therefore possible to make it sink. As a rule, the detection film should float on the surface in an aqueous system.
Publications to familiarise yourself with the subject matter
- Petersen, M., Talledo, S., Bürger H., Bernhard, S., Wilke, T. (2026). Sample without bang: A reusable hydrogen detection foil for school lessons. CHEMKON 33(1), 11-16. doi: https://doi.org/10.1002/ckon.202500029
- Petersen, M., Bauschulte, J., Talledo, S., Hotzel, K., Wark, M., Peneva, K., Bernhard, S., Wilke, T. (2025) Synthesis of Polymeric Carbon Nitrides in a Low-Cost Moka Furnace for Photocatalytic Hydrogen Generation with Visible Light. J. Chem. Educ, 102 (7), 2912-2919. doi: https://doi.org/10.1021/acs.jchemed.5c00114
- Talledo, S., Kubaney, A., Baumer, M.A., Pietrak, K., Bernhard, S. (2024) High throughput methodology for investigating green hydrogen generating processes using colourimetric detection films and machine vision. Digital Discovery, 3 (7), 1430-1440. doi: https://doi.org/10.1039/D4DD00070F



