Navigation
- Home page of the learning unit
- Building block 1: The Dalton atomic model
- Module 2: The order of the elements
- Unit 3: Electrical phenomena
- Unit 4: Excursus - Radioactivity
- Module 5: The Rutherford scattering experiment
- Module 6: In learning stations to the atomic nucleus
- Module 8: Ionisation energies
Module 7.1 Emission spectra
The flame colouring experiment shows you a phenomenon that was already an unexplained phenomenon for natural scientists throughout the 19th century: when elements are excited, they emit light in very specific wavelengths, even if our eyes only have a certain perception of colour, this light is also a mixture of different colours with very specific wavelengths.
The excitation of the elements takes place through the supply of energy. In this case by the thermal energy of the burner flame. In fireworks, the glow is also excited by thermal energy, in fluorescent tubes by electrical energy. You were able to recognise the so-called emission spectra of the elements using the hand spectroscope. The following illustration shows you an overview of the emission spectra of some elements.

The lines that an element shows in the emission spectrum are always the same and they are specific to the respective element. From this we can deduce that the origin of these lines must lie in the structure of the atoms of the element. The atoms of an element must all have something in common that is different from the atoms of other elements.
Rutherford's model of the structure of the atom cannot provide us with an explanation for the origin of the emission spectra. We must therefore extend our model.
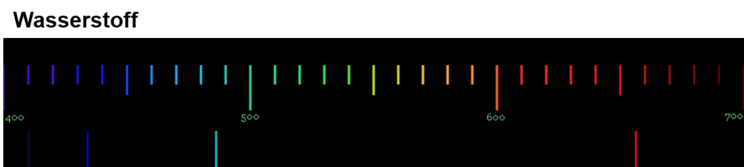
The emission spectrum of hydrogen was of particular interest to the scientists, as it is the element whose atoms have the simplest structure. They therefore hoped that they could explain the cause and laws of light emission using the hydrogen spectrum. The emission spectrum of hydrogen can be generated using a fluorescent tube filled with hydrogen gas. The spectrum of hydrogen consists of 4 lines in the visible range. Their wavelengths are from left to right: 410, 434, 486 and 656 nm.
The following figure shows you the emission spectrum of hydrogen.
Experimental recording of emission spectra
The following pictures show an example of an experimental setup for recording the emission spectrum of hydrogen and the final result. A hydrogen lamp, an optical slit, an optical grating and a simple screen with bleached paper were used. Note that due to the experimental set-up, the longer wavelength red light appears on the left of the screen and not on the right, as above. In addition, the distances between the lines should not be arranged on a scale, as above.
To record the spectrum, it is advisable to completely darken the room and use a camera with automatic exposure time setting. Modern smartphones are also suitable for this.
The Max Planck Institute for Plasma Physics provides instructions for building a mobile phone spectrometer on its website. Mobile phone spectrometer






