Worksheet 1
-
 Worksheet: Experiments with everyday objects On this worksheet you will find the instructions for the experiments that you should carry out at the beginning of this unit.
Worksheet: Experiments with everyday objects On this worksheet you will find the instructions for the experiments that you should carry out at the beginning of this unit.
Module 3: Electrical phenomena
Task 3.1 (introduction)
Before you start:
Summarise: What does our model of atoms look like? What are substances made of?
Task 3.2
At the beginning of this module, you should also carry out a few small experiments with everyday objects. Use worksheet 1 for this.
If you are unable to carry out the experiments at school or at home, take a look at the alternative.
Worksheet 1
-
 Worksheet: Experiments with everyday objects On this worksheet you will find the instructions for the experiments that you should carry out at the beginning of this unit.
Worksheet: Experiments with everyday objects On this worksheet you will find the instructions for the experiments that you should carry out at the beginning of this unit.
Alternative
If you are unable to carry out the experiments at school or at home because you don't have the materials, simply watch the following video as an alternative.
Note: In order to watch the video, you must enter a nickname. You can choose this freely.
If the embedded video is not playable, you can go directly to the video on edpuzzle here
Findings so far
Dalton's hypotheses give us a way of understanding the structure of substances both mentally and with symbols. We can also use this model to explain the laws of chemical reactions.
However, in the experiments with everyday objects, you were able to produce phenomena in a simple way that we cannot explain with Dalton's model: electrical phenomena.
What does a tube television have to do with the building blocks of matter?
At the end of the 19th century, many scientists investigated electricity and electrical phenomena, as not much was yet known about their laws and causes.
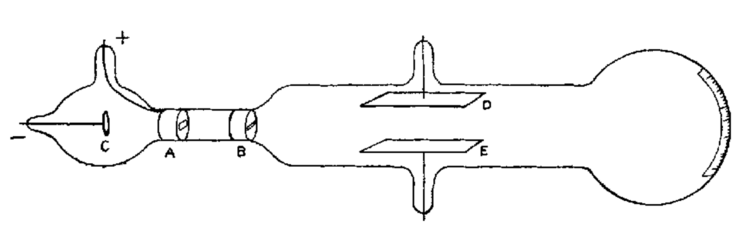
One of these phenomena was cathode rays. They can be generated in a glass tube by applying a high voltage to a metal electrode. The following sketch shows a classic cathode ray tube as used by J. J. Thomson in his experiments at the time.
| C | Cathode | A | Gap (anode) | B | Gap | D/E | Aluminium plate |
The air is sucked out of the entire tube, i.e. it is evacuated. A negative voltage is applied to the cathode (negative pole) and a positive voltage to the anode (positive pole). The beam then passes from the cathode through the two gaps and between the aluminium plates. At the end of the tube it hits a screen and produces a glow. The screen is provided with a scale.
In 1897, Thomson investigated the properties of the cathode rays and found that their path changed when a battery was connected to the aluminium plates so that one plate was positively charged and the other negatively. The cathode ray is then deflected in the direction of the positively charged plate. The more charged the plates are, the greater the deflection of the beam. The beams behave in a similar way when they pass through a magnetic field.
In the following animation, you can take another look at the processes described in Thomson's experiment.
Findings from Thomson's experiments
The deflection in the cathode ray tube shows that the rays behave like negatively charged particles. Thomson was also able to establish that the behaviour of the rays is always the same, regardless of the materials used, and also calculated that these negative particles should be very small and light. With his experiments, Thomson had found a building block of matter that occurs in all atoms: the negatively charged electron.
Questions from Thomson's experiments
Thomson had thus discovered a completely new particle with his experiments: the negatively charged electron. It occurs in all atoms.
Now, of course, the questions arise: How exactly are atoms structured? Where are the electrons located in the atom? How are they arranged?
Application of cathode rays
The properties of cathode rays were used for several decades in televisions and other screens. In order to be able to direct the rays to any point on the screen and generate a corresponding colour glow on the specially coated screen, only two additional plates are needed, which can also deflect the rays to the left and right. This is one of the ways in which electrons have contributed to the spread of television.
For those interested:
This video explains exactly how the tube screens work.