Navigation
- Home page of the learning unit
- Building block 1: The Dalton atomic model
- Module 2: The order of the elements
- Unit 3: Electrical phenomena
- Unit 4: Excursus - Radioactivity
- Module 5: The Rutherford scattering experiment
- Module 6: In learning stations to the atomic nucleus
- Module 7: Light, colour and energy
Module 8: The ionisation energy
In Module 7, you learnt about the energy level and shell model of the electron shell. Balmer and Rydberg made an important contribution to the development of these models with the formula they developed. Balmer and Rydberg's formula could be interpreted using the relationship between the wavelength and energy of light found by Einstein. From this we were first able to develop the energy level model and then the shell model of the electron shell.
In this model, a hydrogen atom is made up of an atomic nucleus with a nuclear charge of +1 and an electron with a charge of -1. The electron is at a fixed energy level, or a shell with a fixed distance from the atomic nucleus.
With the Rydberg formula
\(\frac{1}{\lambda}=R_\infty\cdot\left(\frac{1}{{n_1}^2}-\frac{1}{{n_2}^2}\right)\)
we can determine the wavelength of light emitted by an electron during the transition between 2 energy levels.
Let us now imagine that the electron of the hydrogen atom is to transition from the ground state n₁ = 1 to an energy level n₂, which is virtually infinitely far away from the atomic nucleus. As "infinity" is not a number that we can use in the formula, we simply enter a comparatively very large number for n₂ in the calculation, for example 50. We then obtain 91 nm for the wavelength of the light.
With the help of Einstein's formula:
\(E= \frac{\left(h\cdot c\right)}{\lambda}\)
which describes the relationship between the wavelength and energy of light, we can also calculate how much energy the electron must absorb in order to reach this energy level. We obtain an energy of 13.6 eV.
We have assumed that energy level 50 is almost infinitely far away from the nucleus, but how can we be sure that this is the case? If we carry out the same calculations for the energy level 100, we obtain almost identical values for the wavelength and the energy. They only differ from the previous ones in the 2nd decimal place. The differences are therefore negligible. The electron can absorb a maximum of 13.6 eV of energy. This is the energy required to detach the electron from the attractive force of the positively charged nucleus.
We call this energy the ionisation energy.
Ionisation energies of the elements
With the Rydberg formula, we have a way of calculating the ionisation energy of a hydrogen atom. However, as this formula only describes the hydrogen atom, we cannot apply it to other elements whose atoms have a more complex structure than the hydrogen atom.
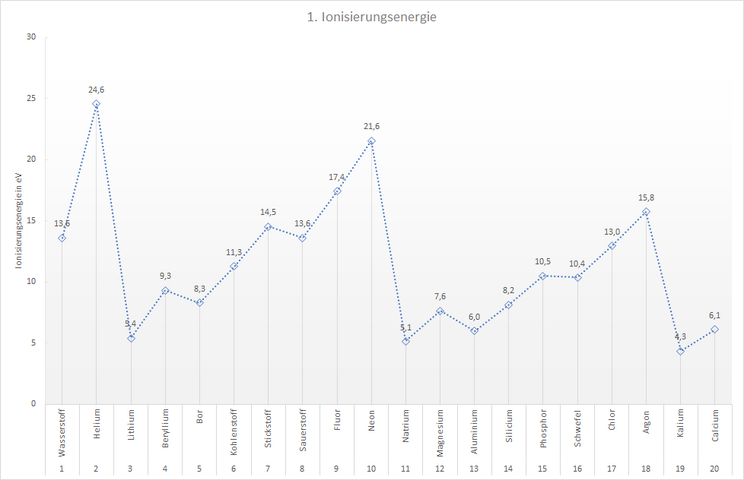
However, the ionisation energy of the elements can be determined with experiments. For example, we can draw up a table showing the minimum amount of energy required to remove exactly one electron from an atom of an element. This 1st ionisation energy is shown for the first 20 elements in the following table.
| Element | Ionisation energy in eV |
|---|---|
| Hydrogen | 13,6 |
| helium | 24,6 |
| Lithium | 5,4 |
| Beryllium | 9,3 |
| boron | 8,3 |
| Carbon | 11,3 |
| Nitrogen | 14,5 |
| Oxygen | 13,6 |
| Fluorine | 17,4 |
| Neon | 21,6 |
| Sodium | 5,1 |
| magnesium | 7,6 |
| Aluminium | 6,0 |
| Silicon | 8,2 |
| Phosphorus | 10,5 |
| Sulphur | 10,4 |
| Chlorine | 13,0 |
| Argon | 15,8 |
| Potassium | 4,3 |
| Calcium | 6,1 |
| ... |
Task
- Use the table to create a diagram that shows the progression of the ionisation energy of the first 20 elements.
- Using the diagram, formulate rules that describe the rough progression of the ionisation energy:
- From top to bottom (within a main group)...
- From left to right (within a period)...
Ionisation energy curve in the periodic table
Your diagram should look something like the example shown here.
You can see that...
- the ionisation energy within a main group decreases from top to bottom. Example:
| Lithium | 5.4 eV |
| Sodium | 5.1 eV |
| Potassium | 4.3 eV |
- the ionisation energy increases overall from left to right within a period. Example:
| Lithium | beryllium | boron | carbon | nitrogen | oxygen | fluorine | neon |
| 5.4 eV | 9.3 eV | 8.3 eV | 11.3 eV | 14.5 eV | 13.6 eV | 17.4 eV | 21.6 eV |
Task
- Use the shell model of the electron shell to explain why...
- ... the ionisation energy decreases so much with the beginning of a new period (example: from neon to sodium).
- ... the ionisation energy within a main group decreases from top to bottom (example: from lithium to potassium).
- ... the ionisation energy increases overall from left to right within a period (example: from lithium to neon).
Particularly stable conditions
You have probably already noticed that the ionisation energy does not increase evenly from left to right within a period, but that there are even elements whose ionisation energy is significantly greater than the trend within the period. Their "successor elements" have a lower ionisation energy.
In the 2nd period these are the elements beryllium and nitrogen, in the 3rd period magnesium and phosphorus. This trend continues in the following periods: the elements of the 2nd main group and those of the 5th main group have significantly higher ionisation energies than would be expected. The electrons are therefore more strongly bound to the atom in these elements than in their "neighbours".
If there are 2 or 5 electrons on the outer shell, it is more difficult to remove an electron from the atom than if the outer shell were occupied by 3 or 6 electrons. For our shell model, this means that there appear to be particularly stable states for the arrangement of the electrons. The shell model cannot provide us with an explanation for this.