Navigation
- Home page of the learning unit
- Building block 1: The Dalton atomic model
- Module 2: The order of the elements
- Unit 3: Electrical phenomena
- Unit 4: Excursus - Radioactivity
- Module 5: The Rutherford scattering experiment
- Module 6: In learning stations to the atomic nucleus
- Module 8: Ionisation energies
Flame colouring worksheet
-
 Flame colouring worksheet On this worksheet you will find the instructions for the flame colouring experiment.
Flame colouring worksheet On this worksheet you will find the instructions for the flame colouring experiment.
Module 7: Light, colour and energy
Module 7 contains four further sections that can be accessed via the links at the bottom of the page or the top navigation bar.
Light and colour
You've probably held a CD or DVD in your hand before, but have you ever taken a close look at it?
Task
Take one of these discs and hold the underside up to the light, turn and rotate it.
Describe your observations.
You can see a natural phenomenon on this DVD that is similar to the rainbow. On a CD or DVD, the light is broken down into its colours by diffraction on the special surface. In the case of a rainbow, this happens through the refraction of light in raindrops.
DVD Oberfläche und Lichtbrechung im Regentropfen
Dieses Bild zeigt dir die Oberfläche einer DVD. Die aus vielen kleinen aufeinanderfolgenden Furchen aufgebaut ist.
Das Bild wurde mit einem Rasterkraftmikroskop aufgenommen.

Dieses Bild zeigt dir, wie weißes Sonnenlicht in einem Regentropfen gebrochen wird, sodass man alle Spektralfarben in einem Regenbogen sehen kann.
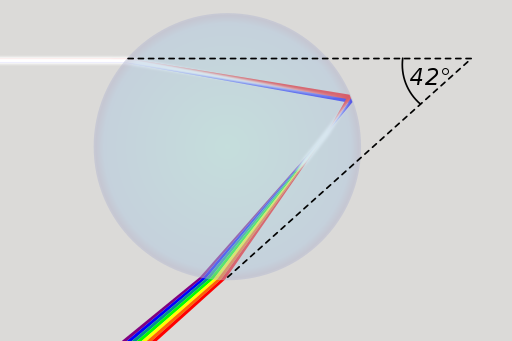
These phenomena show us one thing above all: sunlight as we know it is not simply "white light", but is made up of all the colours of the colour spectrum. We can characterise light physically and mathematically as a wave and describe the colours that we perceive using their respective wavelengths. The visible range of light extends from violet light with a wavelength of around 400 nm to red light, which has a wavelength of up to 750 nm. Below 400 nm is the range of ultraviolet light (UV radiation), above around 750 nm is the range of infrared light.
However, you are probably also familiar with light sources that do not emit white light. Two examples of this are fluorescent tubes, such as neon tubes, or fireworks, which can glow in many different colours.
Task
How can we investigate whether the light from such light sources is also composed of different colours?
To do this the experiment: Flame colouring. If you can't carry out the experiment at school, watch the video on the experiment as an alternative.
Then complete the following tasks:
-
Describe your observations when you look at the flame without and with a hand spectroscope.
-
Which element of the different salts cause the different colours of the flame? Explain your answer.
- Can you explain these observations using Rutherford's core-shell model?




