Navigation
- Home page of the learning unit
- Building block 1: The Dalton atomic model
- Module 2: The order of the elements
- Unit 3: Electrical phenomena
- Unit 4: Excursus - Radioactivity
- Module 5: The Rutherford scattering experiment
- Module 6: In learning stations to the atomic nucleus
- Module 8: Ionisation energies
Module 7.4 The shell model
You have learnt in the last building blocks that the electrons in the atomic shell can take on different energy levels by absorbing energy and can return to lower energy levels by releasing energy. This process is the cause of the emission of the specific line spectra of the elements.
We can now imagine that there are various permitted states for the electrons in the atomic shell, but the question remains as to how exactly the electrons of an atom are distributed there.
The physicist Niels Bohr also began working on this question in 1913. He developed a model for the structure of atoms that is known today as Bohr's atomic model or shell model.
He calculated important properties of the energy levels of the electrons and used the periodic properties of the elements to determine the distribution of the electrons on the energy levels.
The following table shows the distribution of electrons on the energy levels for the first 18 elements:
Electrons at energy level n = 1 | Electrons at energy level n = 2 | Electrons at energy level n = 3 | |
1 H - hydrogen | 1 | ||
2 He - helium | 2 | ||
3 Li - Lithium | 2 | 1 | |
4 Be - Beryllium | 2 | 2 | |
5 B - boron | 2 | 3 | |
6 C - Carbon | 2 | 4 | |
7 N - Nitrogen | 2 | 5 | |
8 O - Oxygen | 2 | 6 | |
9 F - Fluorine | 2 | 7 | |
10 Ne - Neon | 2 | 8 | |
11 Na - sodium | 2 | 8 | 1 |
12 Mg - magnesium | 2 | 8 | 2 |
13 Al - aluminium | 2 | 8 | 3 |
14 Si - Silicon | 2 | 8 | 4 |
15 P - Phosphorus | 2 | 8 | 5 |
16 S - Sulphur | 2 | 8 | 6 |
17 Cl - chlorine | 2 | 8 | 7 |
18 Ar - argon | 2 | 8 | 8 |
Task
- Use the electron distribution table to explain why the elements of the main groups are similar in their chemical behaviour.
- Explain why the noble gases have a special position in the periodic table.
- According to this principle, how many electrons would be at the next energy level n = 4 ?
Chemical behaviour and electron distribution from the 4th period onwards
The elements in a main group behave so similarly chemically because they always have the same number of electrons at their highest energy level. These determine the reaction behaviour of the elements.
The noble gases occupy a special position here, their uppermost energy level is fully occupied and therefore particularly stable. They therefore only react with other elements with great difficulty.
At the next energy level n = 4, 18 electrons can be present for the elements potassium to krypton (19 to 36) according to the above diagram.
However, this means that the elements from gallium to krypton (31 to 36) would not have the same number of electrons at their highest energy level as their related elements in periods 1 to 3.
Bohr solved this problem by introducing sub-stages. The electrons of elements 21 to 30 are therefore on their own energy level, on which 10 electrons can reside. Only when this sub-level is full is the energy level n = 4 filled up further. This can be illustrated using an example:
The element arsenic has 33 electrons in its shell. The first 18 electrons are in the first three energy levels, as shown in the table. Electrons 19 and 20 are at energy level n = 4. The 21st electron is bound to a sublevel, as are the 22nd, 23rd and so on until the sublevel is filled with 10 electrons. From the 31st electron, the energy level n = 4 is then filled up again until there are a total of 5 electrons on it, so that there are also 5 electrons on the top energy level in the element arsenic. This lower level, introduced by Bohr, could be called n = 4a or n = 4.1.
Bowls as energy levels
You have now learnt how the electrons are distributed across the energy levels in the atomic shell. However, the question remains as to how we can visualise these energy levels. Bohr also tried to answer this question.
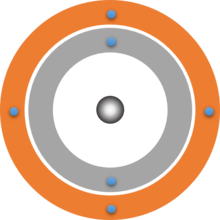
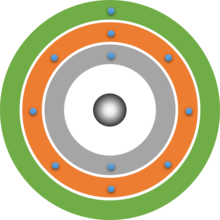
Dalton's atomic model was already based on the idea that atoms are spherical. Bohr was now even able to calculate that the electrons are at a certain distance from the atomic nucleus. So-called shells are formed around the atomic nucleus. This model can be easily combined with the model of energy levels. The shell closest to the nucleus corresponds to the lowest energy level n = 1. 2 electrons can be accommodated on this shell. The next shell is further away from the nucleus and can hold up to 8 electrons, this is the energy level n = 2.
In this way, we can gradually build up a two-dimensional model of an atom consisting of a nucleus and a shell of electrons.
Task
- Using the electron distribution table and the information from the previous text, draw a shell model of an atom of the following elements:
- Hydrogen
- helium
- carbon
- oxygen
- sodium
- chlorine
Shell models of atoms
Your drawn shell models should look something like the following three examples. Note that the atomic nuclei are only represented here by simple grey spheres. The exact structure of the atomic nucleus is not shown here. It is dealt with in Building Block 6.