Module 5: The Rutherford scatter test
Radioactive radiation as an examination tool
The novel phenomenon of radioactivity was a promising field of research for many scientists at the beginning of the 20th century, including Ernest Rutherford (1871-1937). He was a New Zealand physicist and was awarded the Nobel Prize in Chemistry in 1908 "for his investigations into the decay of the elements and the chemistry of radioactive substances". Like many scientists at the time, he also wanted to find out how atoms are constructed.
From 1907, he worked as a professor at the University of Manchester and, together with his colleagues, investigated the properties of a certain part of radioactive radiation. This part is called α-radiation. It consists of two positively charged helium atoms, the α particles. They can penetrate thin layers of other materials. In 1908, his colleague Hans Geiger discovered that a narrow beam of α particles appears to fan out when it hits or penetrates a thin gold foil. The scattering that Geiger was able to determine was in the range of approximately one degree. Rutherford then entrusted Geiger and another young colleague, Ernest Marsden, with the task of investigating this scattering in more detail. Rutherford wanted to find out whether they could also be backscattered. But he didn't think it was possible.
Geiger and Marsden therefore designed an experiment to investigate whether the α-particles could be scattered by a thin metal foil by more than just one degree. In the years from 1908 to 1913, they were able to show through several extensive experiments that α-particles are scattered more strongly by matter than was generally assumed.
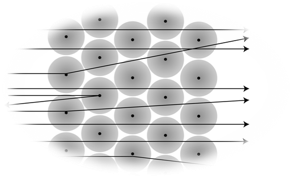
You can see one of the most important experiments in the following animation.
Task 5.1
Click through the animation and summarise the information in your own drawing of the experiment so that you can solve the digital task afterwards.
Tasks for animating the scatter test
Model test
Task 5.3
We can't carry out Rutherford's scattering experiment ourselves, so we use a model experiment to simulate it. For this we need a shoe box, some cardboard, small metal balls and nails.
If you are unable to carry out the experiment yourself at school (or at home), simply watch the following video.
- Watch the video carefully and write down your observations.
- Based on your observations, make assumptions about what might be under the cardboard lid.
The Rutherford scattering experiment in retrospect
Rutherford himself later summarised the research of his working group in retrospect. In order to better describe the results of the experiment and thus the structure of the atoms, he and his team had to make some assumptions.
Task 5.4
Read the texts in the file that you will find under "Material". You can choose between a German translation or the original in English with translation aids.
Mark important statements and assumptions about the structure of atoms and summarise them in writing.
The model of an atom designed by Rutherford
Task 5.5
Complete the following tasks.
- Using the information from the previous building blocks, the animation, the model experiment and the assumptions made by Rutherford, develop your own mental model of an atom. Draw your model.
- Name properties that you can make about the structure of the atom based on Rutherford's model.
- Evaluate Rutherford's model. Which properties of an atom are not described in the model?
If you get stuck, the tips below may help you.
Tips
Model test part 2
Now we come to the resolution of the question from the pilot project. Watch the following video again carefully.
Explanations on the pilot project
As you saw at the end of the video, our model test is not an ideal representation of the real scatter test.
There are a few things to consider here:
- In the model experiment, we only used a single row of nails. Although the gold foil in the real experiment was very thin, it was still about 1000 atoms thick.
- The size ratios between metal spheres and nails are completely different to those between the alpha particles and the atomic nuclei of the gold.
- In our model, the spheres are larger than the nails. In reality, the alpha particles are much smaller than the atomic nuclei of gold.
- And: the distances between the atomic nuclei (the nails) are much too small.
- In the model experiment, the spheres are deflected when they hit the nail; in the real experiment, the alpha particles were deflected simply because they came close to the nucleus, because both are positively charged and like charges repel each other.
Nevertheless, we can use this simple model to illustrate important parts, properties and results of the real experiment.