Fukushima
Fukushima
Contents
- Measurement data on radioactivity in the air in Oldenburg
- Measurement data on radioactivity in other samples from Oldenburg
- Measurement data on radioactivity in the air in Germany and worldwide
- Data on radiation dose in the immediate vicinity of the plant
- Information from external institutions on the reactor accident in Fukushima
- Radiation dose limits in Germany
- Health consequences of increased radiation dose
- Explanation of technical terms
Measurement data on radioactivity in the air in Oldenburg
The Institute of Physics at the University of Oldenburg continuously measured the radioactivity in the air at the Wechloy site from 29 March 2011 to 9 May 2011. Random measurements were then carried out. During the continuous measurement period, traces of iodine-131 (J-131), caesium-137 (Cs-137) and caesium-134 (Cs-134) were detected, which very probably originated from the destroyed nuclear reactors in Fukushima (Japan). Iodine-131, caesium-137 and caesium-134 do not occur in nature. For the measurement, air was sucked through a glass fibre filter over a collection period of several hours, which was then analysed using gamma spectrometry. The following graph shows the concentration of iodine-131 in mBq/m³(millibecquerel per cubic metre = thousandth of a becquerel per cubic metre) averaged over the respective collection period.
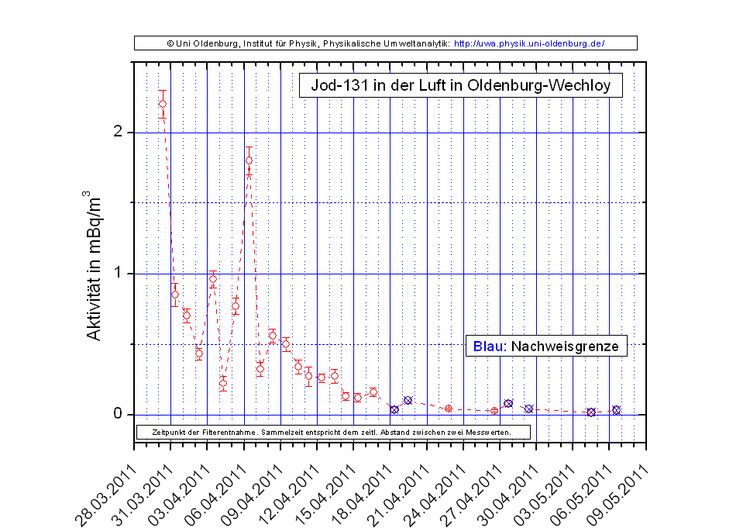
An adult breathes in about (10 - 12) m³ of air per day. Even if all of the iodine-131 from the air we breathe remained in the body, only approx. (1 - 25) mBq of iodine-131 would enter the body per day at the stated concentrations. There is no health risk associated with this. For comparison: the measured iodine-131 concentrations are at least approx. 1,000 times lower than the typical concentration of natural radioactivity in the air. To detect caesium in the air, the air filters for the collection period from 29.03.11 to 10.04.11 were analysed together using gamma spectrometry. The volume of air drawn through the filters during this period was approx. 4,200 m³. In this volume, (0.044 ± 0.004) mBq/m³ Cs-137 and (0.041 ± 0.002) mBq/m³ Cs-134 were detected. The activity ratio of both isotopes of approx. 1:1 corresponds to the information provided by the operator of the destroyed plants(TEPCO) and is therefore an important indication of the origin of the iodine and caesium from Fukushima. The measured caesium concentrations are also so low that there is no health risk associated with them.
Measurement data on radioactivity in other samples from Oldenburg
- Pasture grass: An iodine-131 concentration of (2.9 ± 0.5) becquerels per kilogramme of fresh material was detected in a grass sample from a pasture in Oldenburg-Wechloy at the beginning of April 2011. The grass sample came from a pasture area of approx. 2 m². Comparable concentrations were also measured at other locations.
Measurement data on radioactivity in the air in Germany and worldwide
The Federal Office for Radiation Protection (BfS) measures radioactivity in the air at its Schauinsland measuring station for atmospheric radioactivity. Traces of iodine-131 and caesium-137 have been detected in the air there since 25 March 2011. The values were in the same order of magnitude as those in Oldenburg.
In addition, the BfS also publishes measurement data from other institutions:
- the Physikalisch Technische Bundesanstalt (PTB) in Braunschweig and
- the German Weather Service (DWD) in Offenbach and Potsdam.
The highest value for iodine-131 from Fukushima in Germany was measured in Braunschweig at 3.7 mBq/m³.
All iodine-131 concentrations are at least approx. 1,000 times lower than the typical concentration of natural radioactivity in the air. There is no health risk associated with these concentrations.
How and within how long the radioactivity spread from Fukushima to Germany can be tracked using measurement data from the InternationalComprehensive Nuclear-Test-Ban Treaty Organisation(CTBTO).
Data from 02.04.11
According to the operator(TEPCO), a crack about 20 cm long has appeared in a cable shaft, from which highly radioactive water is flowing directly into the sea. Radiation dose rates of over 1,000 mSv/h were measured inside the shaft and radiation dose rates of 400 mSv/h outside the shaft. These values represent a massive health hazard.
A radiation dose of approx. 4.5 Sv (= 4,500 mSv) leads to the death of 50% of the people affected within a month without medical intervention. With a radiation dose rate of 400 mSv/h, this is reached in approx. 11 hours. A radiation dose from approx. (6 - 8) Sv is fatal for 100 % of those affected.
The International Atomic Energy Agency IAEA has measured radiation dose rates of (0.6 - 4.5) μSv/h at 7 measuring points north and north-west of the reactors at a distance of (32 - 62) km. These values are approximately (7 - 55) times higher than the values of the natural radiation dose rate.
Data from 17/03/11
According to the operator(TEPCO), radiation dose rates of 400 mSv/h were measured at 10 am on the landward side of unit 3 and 100 mSv/h on the landward side of unit 4. These values represent a massive health hazard.
Data from 16/03/11
According to the operator(TEPCO), dose rates of 8 mSv/h (0 o'clock) and 11 mSv/h (12 o'clock) were temporarily measured at the main gate of the plant.
Data from 15/03/11
The International Atomic Energy Agency (IAEA ) reports that after another explosion and the outbreak of a fire, the radiation dose rate at certain locations in the plant has reached 400 mSv/h (millisieverts per hour).
The IAEA later reports that the dose rate of 400 mSv/h was only measured at a limited location in the plant at a specific time. In the meantime, dose rates of 11.9 mSv/h (01.00) and 0.6 mSv/h (07.00) were measured.
Radiation dose limits in Germany
- The natural radiation dose in northern Germany is approx. 2.4 mSv per year (see → Radiation dose rate). Of this, around 0.7 mSv per year is due to external radiation (radiation from space and radiation from naturally occurring radioactive substances). The remaining radiation dose is caused by naturally occurring radioactive substances that are absorbed into the body with food or the air we breathe.
- A dose rate of 0.7 mSv per year corresponds to approx. 0.00008 mSv/h (millisievert per hour) = 0.08 μSv/h (microsievert per hour).
- According to the Radiation Protection Ordinance, the radiation dose to the population from the normal operation of nuclear facilities in Germany may not exceed 0.3 mSv per year. In the worst-case scenario, the total dose must not exceed 50 mSv.
- For persons with academic appointments, the radiation dose must not exceed 20 mSv per year. In individual cases, the competent authority may authorise a dose of 50 mSv per year, whereby the total dose may not exceed 100 mSv for five consecutive years. In addition, the dose may not exceed 400 mSv over the entire working life.
Health consequences of increased radiation dose
- Every additional dose of radiation adversely affects human health. Even low radiation doses increase the risk of subsequent radiation-induced cancer ("stochastic radiation damage"). The higher the dose, the greater the risk.
- There is no threshold value below which a radiation dose is harmless.
- High radiation doses (from approx. 500 mSv), which are absorbed within a short period of time, lead to acute radiation damage such as nausea, fatigue, changes in the blood count, etc. within hours, days or weeks ("deterministic radiation damage").
- A radiation dose of approx. 4.5 Sv (= 4,500 mSv) leads to death within a month in 50% of the people affected without medical intervention. A dose of approx. 6 - 8 Sv is fatal for 100 % of those affected.
Explanation of technical terms
- Becquerel: Becquerel (Bq) is the unit of radioactivity. It indicates how many atomic nuclei of a substance (e.g. iodine-131) decay per second. For example: 100 becquerels = 100 Bq means that 100 radioactive decays take place every second. Radioactive radiation is therefore released 100 times per second. Only after the physical half-life has elapsed has the radioactivity fallen to half of the original value, in this example to 50 radioactive decays per second.
The activity is often specified in smaller units, e.g: - Millibecquerel (mBq): 1 mBq = 1 thousandth of a becquerel.
- Caesium-134/137: Caesium-137 (Cs-137) and caesium-134 (Cs-134) are radioactive substances that do not occur in nature. They are produced by nuclear fission in nuclear reactors. Their physical half-lives are 30 years (Cs-137) and 2.06 years (Cs-134). During radioactive decay, caesium-137 is converted into barium-137 and caesium-134 (almost 100 %) into barium-134. Beta and gamma radiation is released in the process.
Caesium enters the body through food and is stored in muscle tissue. Its biological half-life is approx. (110 - 140) days. - Half-life: The physical half-life indicates the time within which the radioactivity of a substance has reduced to half its original value. Examples: for iodine-131 the physical half-life is approx. 8 days, for caesium-137 approx. 30 years, for strontium-90 approx. 29 years and for plutonium-239 approx. 24,000 years.
The biological half-life indicates the time it takes for the amount of a substance absorbed by the body to be reduced by half through excretion.
The effective half-life indicates the time it takes for the amount of a radioactive substance absorbed by the body to be halved through excretion and radioactive decay - Iodine-131: Iodine-131 (J-131) is a radioactive substance that does not occur in nature. It is produced by nuclear fission in nuclear reactors. Its physical half-life is 8.02 days. During radioactive decay, iodine-131 is converted into the noble gas xenon-131 (Xe-131). Beta and gamma radiation is released in the process.
Iodine-131 enters the human body through respiration and food and is primarily stored in the thyroid gland. There it can cause thyroid cancer if present in large quantities. The biological half-life of iodine-131 is approx. 80 days. - Sievert: Sievert (Sv) is the unit of → radiation dose.
- Radiation dose: The radiation dose ("effective dose") is measured in the unit sievert (Sv). It indicates how much energy from radioactive radiation has been absorbed by the body. The radiation dose is the quotient of the absorbed radiation energy (measured in joules (J)) and mass (measured in kilograms (kg)). This quotient has been given the name "Sievert". The following applies:
1 Sievert = 1 Joule per kilogramme or, in abbreviated form, 1 Sv = 1 J/kg
An example: when irradiated with gamma radiation from outside, a radiation dose of 1 Sv means that gamma radiation with a total energy of 1 Joule was absorbed per kilogramme of body mass. For example, 1 joule corresponds to the energy of gamma radiation from approx. 20,000 billion = 20 trillion radioactive decays of iodine-131 (J-131).
The radiation dose is often given in smaller units, e.g: - Millisievert (mSv): 1 mSv = 1 thousandth of a sievert,
- Microsievert (μSv): 1 μSv = 1 millionth of a sievert,
- Nanosievert (nSv): 1 nSv = 1 billionth of a sievert.
- Radiationdose rate: The radiation dose rate is the radiation dose per time. It is given in units of sievert per hour (Sv/h), millisievert per hour (mSv/h), sievert per year (Sv/a), etc.
- Radiation damage: in the case of "stochastic" (random) radiation damage, the probability of the disease occurring depends on the radiation dose level, but not the severity of the disease (e.g. cancer). In the case of "deterministic" (predetermined) radiation damage, on the other hand, the severity of the disease (e.g. skin burns, diarrhoea, weight loss) increases with a higher dose.
