Kontakt
Cryptochromes
Cryptochromes are evolutionarily related to photolyases, which are an ancient family of DNA-binding proteins that can absorb blue-light via a bound flavin molecule. Cryptochromes are key regulators of circadian processes, but some isoforms of cryptochromes are currently investigated as primary receptors mediating avian magnetoreception. Expression of cryptochrome 4 (Cry4) in European Robin in double cones and long wavelength single cones suggests involvement of photoreceptor specific proteins in Cry4 signaling. A search for Cry4 interaction partners has identified so far six candidates that are expressed in the outer or inner segments of photoreceptor cells.
These proteins are long wavelength-sensitive opsin (also called red opsin or iodopsin), which is the visual pigment present in the long wavelength sensitive single cones and in the double cones. The screening resulted further in the α- and γ-subunit of the cone specific variant of a heterotrimeric G protein (Gtα and Gtγ), two components of the visual cycle (the retinol binding protein RBP and the retinal G protein-coupled receptor (RGR), a non-visual opsin). Finally, one candidate is a subunit of voltage-gated potassium channel (Kv8.2).
Therefore, Cry4 could trigger or participate in photoreceptor signaling. Different scenarios of intracellular Cry4 signaling are conceivable, but await further experimental verification.
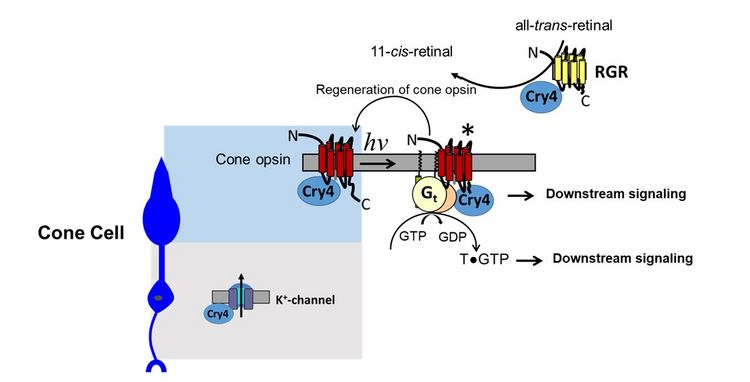
Figure 1: Different intracellular signaling scenarios involving putative Cry4 interacting proteins.
LITERATUR
Günther, A., Einwich, A., Sjulstok, E., Feederle, R., Bolte, P., Koch, K.-W., Solov’yov, I.A. and Mouritsen, H., (2018) Double-cone localisation and seasonal expression pattern suggest a role in magnetoreception for European robin cryptochrome 4. Curr. Biol. 28, 1-13.
Wu, H., Scholten, A., Einwich, A., Mouritsen, H., and Koch, K.-W. (2020) Protein-protein Interaction of the Putative Magnetoreceptor Cryptochrome 4 Expressed in the Avian Retina. Sci. Rep. 10(1):7364.
Görtemaker, K., Yee, C., Bartölke, R., Behrmann, H., Voß, J.O., Schmidt, J., Xu, J., Solovyeva, V., Leberecht, B., Behrmann, E., Mouritsen, H., Koch, K.W. (2022) Direct Interaction of Avian Cryptochrome 4 with a Cone Specific G-Protein. Cells 11(13):2043.

