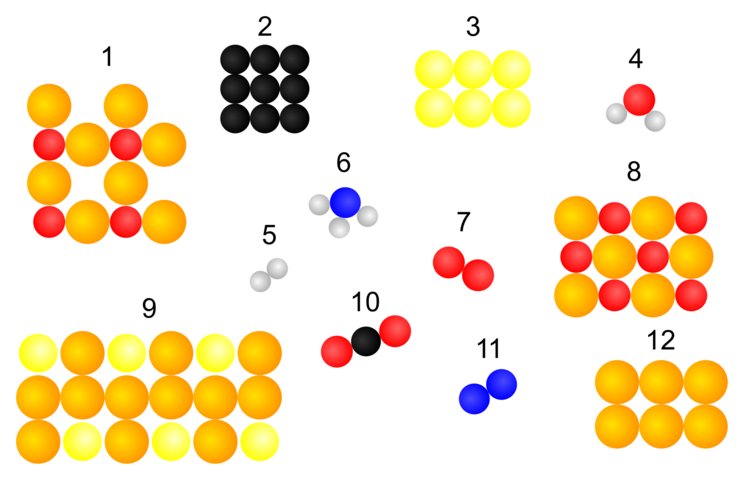
Module 1: The Dalton atomic model
Historical representation
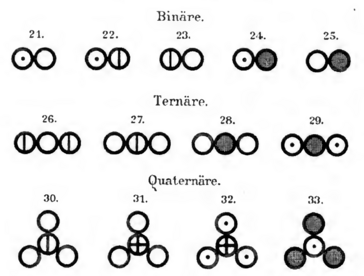
The following two illustrations show tables by John Dalton from 1808. With the help of these illustrations, he introduced a model with which elements and compounds could be represented.
This is why the model on which the representation of the elements and compounds at the top of this page is based is also named after him: Dalton's atomic model.
Task 1.2
- Compare Dalton's representation of the elements and compounds with those at the top of this page.
- Name and explain the similarities and differences in the presentation.
Dalton's hypotheses
At the beginning of the 19th century, Dalton hypothesised in his work "A New System of Chemical Philosophy", based on his investigations into the properties of various gases, that every substance consists of the smallest, indivisible particles, the atoms. Based on this, he made further assumptions:
- The atoms of an element are equal in size and mass; atoms of different elements differ in size and mass.
- Atoms cannot be divided, created or destroyed.
- In a chemical reaction, the atoms of the starting materials are rearranged and combined in certain ratios.
Various phenomena can be explained on the basis of Dalton's assumptions. He was also looking for a unit for atomic mass. He knew that in his time there was no way to determine the actual masses of atoms. They were far too small.
Dalton therefore chose the mass of the lightest atom - the hydrogen atom - as the unit. He gave this atomic mass the value 1, which he then used to compare the masses of the other atoms and arrived at the results that you can find in the supplementary material in the left-hand column. These values are known as relative atomic masses. They do not represent the actual atomic masses, but only the atomic masses in comparison with the mass of a hydrogen atom. He was one of the first scientists to create a table of relative atomic masses based on experimental results. Today's atomic mass unit unit(u) was therefore previously also referred to as the dalton (Da). A hydrogen atom has a relative atomic mass of 1 u.
Task 1.3
With the help of Dalton's atomic model, we can explain and utilise some phenomena of chemical reactions.
- Explain the following observations in your own words, with reference to Dalton's assumptions:
- In a chemical reaction, the total mass of the substances involved is conserved.
- If 2 or more elements combine to form a specific compound, this always happens in a specific, constant mass ratio.
- Example: 88.81 g of oxygen always react with 11.19 g of hydrogen to form 100 g of water.
- Calculate the mass of carbon dioxide produced when 24 g of carbon are completely burnt.