Station 2: Isotopes
The structure of the atomic nucleus
What you took away from Station 1:
The nucleus of an atom is made up of the single positively charged protons and the uncharged neutrons. Both particles have a mass of almost exactly 1 u.
As an atom is electrically neutral to the outside (i.e. uncharged), there are always as many electrons in the atomic shell as protons in the nucleus. An electron has such a small mass that almost the entire mass of an atom is determined by the protons and neutrons.
For example, the nucleus of a gold atom is made up of 79 protons and 118 neutrons. The mass of this atomic nucleus is therefore 197 u. The 79 electrons in the shell together weigh only about 0.04 u.
How can we explain that the atomic mass of the element chlorine is 35.45 u if the mass is only determined by protons and neutrons and these weigh 1 u?
We will return to this question later in this station and you will be able to answer it then.
One element, different masses
At the beginning of the 20th century, scientists discovered something strange through research into radioactive radiation and radioactive elements: there were atoms that belonged to the same element, i.e. they behaved exactly the same chemically, but had different atomic masses.
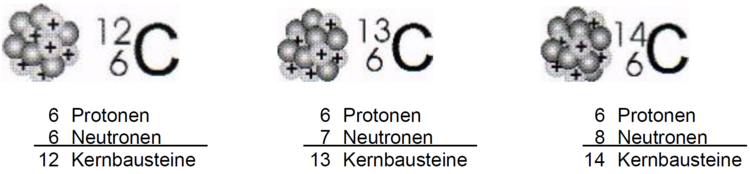
This was first discovered with very heavy elements such as uranium. Today we also know, for example, that almost all carbon atoms have a mass of 12 u, but a few carbon atoms have a mass of 13 u or 14 u. However, all carbon atoms contain 6 proton atoms. However, all carbon atoms contain 6 protons in their nucleus and 6 electrons in the atomic shell. However, their nuclei can contain different numbers of neutrons: 6, 7 or even 8.
Isotopes
These types of atoms, whose atomic nuclei have the same number of protons but different numbers of neutrons, are known as isotopes.
However, isotopes are not only found in heavy radioactive elements or carbon. Mass spectrometry has shown the existence of isotopes in many different elements. Other naturally occurring elements, such as chlorine or neon, also consist of atoms whose atomic nuclei have different masses.
The so-called isotope ratio indicates the relative abundance of the isotopes of an element.
An example: the atoms of the element silver occur in nature with the masses 107 u and 109 u. The nuclei of the atoms differ by 2 neutrons more or less. If you had 100 atoms of silver and could weigh them, 52 of them would weigh 107 u and 48 atoms would weigh 109 u. The relative frequency is 52 % to 48 %. This ratio is always the same.
Task 6.2.1
- Explain the term "isotope" in your own words. Use the illustration of carbon isotopes below.
- All atomic nuclei of the element chlorine contain 17 protons. The atomic mass in the periodic table is given as 35.45 u. Explain how this atomic mass can come about.
- "All atoms of an element are the same." Critically evaluate this statement. Develop a more precise statement.