Station 1: Other building blocks of atoms
Rutherford discovers another building block of the atom
In 1919, Ernest Rutherford researched the interaction of α-particles with various substances. α-particles are the particles that make up radioactive α-radiation (see Building Block 4).
In one experiment, he bombarded gaseous nitrogen with α-particles and, with the help of a special screen, was able to observe that a single positively charged particle was created.
Rutherford surmised that these particles were knocked out of the nitrogen atoms by the α particles. With these experiments, he had found a building block of atoms.
He called this particle, with a mass of 1 u and a single positive charge, a proton (from the ancient Greek 'the first').
In addition to electrons, protons are another building block of an atom. As they are positively charged, they ensure that an atom is electrically neutral overall. A neutral atom is therefore made up of just as many protons as electrons.
We now know that an atom consists of a nucleus and a shell and that the protons are located in the nucleus. The electrons are distributed in the shell of the atom. As the protons in the nucleus are much heavier than the electrons in the shell, most of the mass of an atom is concentrated in the nucleus. But how is it that the protons in the nucleus do not repel each other? They all carry a positive charge and like charges repel each other.
A model experiment on the structure of the atomic nucleus
Task 6.1.1
Place a few identical ring magnets on a firm surface with the same pole facing upwards. Now try to bring the magnets as close together as possible without holding them or placing them on top of each other.
- Describe and write down your observations.
Now place cent coins on the base between the magnets and see how close you can bring them together.
- Describe and write down your observations.
- Interpret the difference between the two experiments. Explain the role of the cent coins in this model experiment.
If you are unable to carry out the experiment yourself, simply watch the following video and complete the tasks.
Another core component
In the model experiment, you were able to feel for yourself that the same charges repel each other using the magnets, which are a model for the protons here. However, if we place coins between the "protons", we can build a "nucleus" that holds together.
It was not until 1932 that James Chadwick, a colleague of Rutherford, was able to provide proof that such particles exist in the atomic nucleus, which act like the coins in the model experiment.
He discovered that the atomic nucleus is not only made up of protons. Neutrons are another building block of the nucleus. In the model experiment, you have already seen the role that neutrons play in the structure of the atomic nucleus. They ensure that the protons can remain close together and that the atomic nucleus remains stable. Neutrons carry no charge (hence the name) and are about as heavy as protons. Both have a mass of 1 u.
Protons and neutrons together make up almost the entire mass of the atom. The mass of an atom is the sum of the protons and neutrons. The very light electrons have almost no influence on the mass of an atom.
Proton number and mass in the periodic table
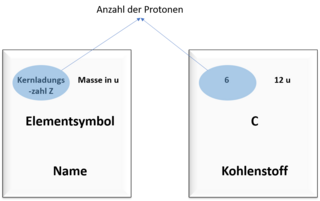
You probably know the periodic table from your lessons. It summarises a lot of information about the elements.
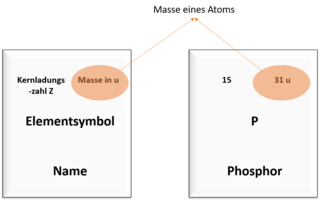
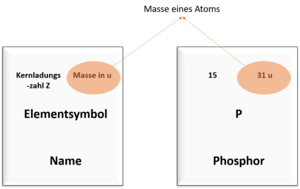
Among other things, you can also read off the number of protons in an atom and the mass of the atom in the periodic table. The pictures show you where you can find which value.
Exercise
Task 6.1.2
The number of neutrons in an atom is therefore the mass number of the atom minus the number of protons in the atom.
An example:
An atom of the element phosphorus has a mass number of 31 and a nuclear charge number of 15.
The number of neutrons is therefore: 31 - 15 = 16
Check what you have learnt in the following exercise.
Task 6.1.3
Summarise the information at this station by answering the following questions:
- Name the particles that make up the atomic nucleus.
- Describe the role of the individual building blocks of the atomic nucleus and name their properties.
- Describe how the mass of an atom is made up. Also assess the influence of the electrons.
How do you imagine an atomic nucleus? Develop a mental model and draw a carbon atom nucleus with 6 neutrons.
Hint: Carbon has a nuclear charge of 6.