Module 7.3: The shell model of the atomic shell
You have learnt in the previous sections that the electrons in the atomic shell differ in their energy content and are at different energy levels. These energy levels are firmly defined and each element has very specific energy levels.
There are therefore different permitted levels for the electrons in the atomic shell, but the question remains as to how we can visualise these levels and how exactly the electrons of an atom are distributed there.
In 1913, the physicist Niels Bohr developed an atomic model that is known today as the Bohr atomic model or shell model.
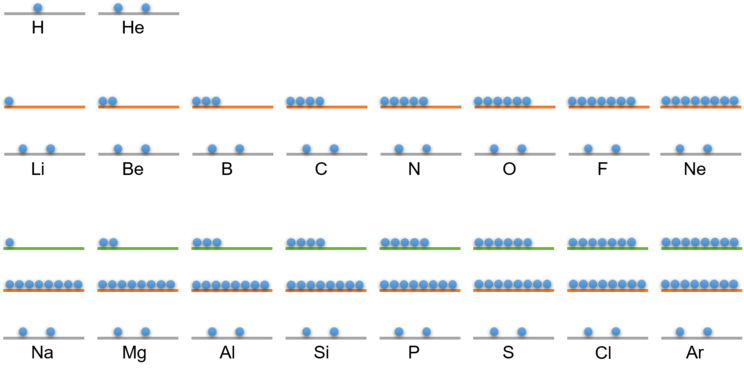
He was able to calculate the distribution of electrons on the energy levels. Each energy level can only hold a certain maximum number of electrons. The following figure shows the distribution of electrons on the first 3 energy levels as determined by Bohr. Below you will also find a table showing the distribution. How the electrons are distributed at higher energy levels is explained in the section below for those interested.
Table of electron distribution
The following table shows you the distribution of electrons on the energy levels for the first 18 elements:
Electrons at energy level 1 | Electrons at energy level 2 | Electrons at energy level 3 | |
1 H - hydrogen | 1 | ||
2 He - Helium | 2 | ||
3 Li - Lithium | 2 | 1 | |
4 Be - Beryllium | 2 | 2 | |
5 B - boron | 2 | 3 | |
6 C - Carbon | 2 | 4 | |
7 N - Nitrogen | 2 | 5 | |
8 O - Oxygen | 2 | 6 | |
9 F - Fluorine | 2 | 7 | |
10 Ne - Neon | 2 | 8 | |
11 Na - sodium | 2 | 8 | 1 |
12 Mg - magnesium | 2 | 8 | 2 |
13 Al - aluminium | 2 | 8 | 3 |
14 Si - Silicon | 2 | 8 | 4 |
15 P - Phosphorus | 2 | 8 | 5 |
16 S - Sulphur | 2 | 8 | 6 |
17 Cl - chlorine | 2 | 8 | 7 |
18 Ar - argon | 2 | 8 | 8 |
Bowls as energy levels
However, the question of how we can imagine these energy levels remains unanswered. Bohr also tried to answer this question.
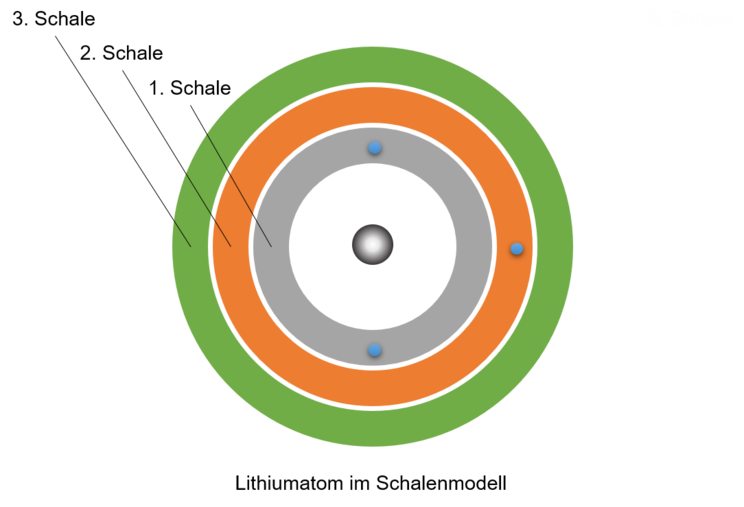
Dalton's atomic model was already based on the idea that atoms are spherical. Bohr was even able to calculate that the electrons must be at a fixed distance from the atomic nucleus. He interpreted this result to mean that the electrons are located in so-called shells around the atomic nucleus. This finding can be easily linked to the model of energy levels. The shell closest to the atomic nucleus corresponds to the lowest energy level. There is room for 2 electrons in this shell. The next shell is further away from the nucleus and can hold up to 8 electrons, this is the second energy level, and so on.
In this way, we can gradually build up a two-dimensional model of an atom consisting of a nucleus and a shell of electrons. The electrons are located in shells. Each shell is an energy level.
In this model, we can imagine the atom as a sphere made up of the small but heavy atomic nucleus and the atomic shell. The atomic shell is made up of the shells. They are located like layers around the atomic nucleus and represent the areas in which the electrons are located.
Task 7.3.1
- Match the drawings of the shell model of an atom to the correct element.
- Using the electron distribution table or the picture of the electron distribution on the energy levels, draw a shell model of an atom of the following elements:
- Hydrogen
- carbon
- oxygen
- lithium
- chlorine
- Potassium *
*You may have to think a little about this task.
Explanation of flame colouring with the shell model
Task 7.3.2
We have starting from the problem of how the lines of the emission spectra can be explained, we worked outhow the shell of an atom is constructed. You should now link this knowledge.
- Summarise in your own words how the formation of the emission lines can be explained using the shell model.
- Then solve the digital task by filling in the gaps in the text with the appropriate terms. This will give you a sample solution for your summary.
Task 7.3.3
Lithium compounds, like elemental lithium, show a red flame colour. You can look at the lines of the emission spectrum again in Building Block 7.1. You are now to trace the formation of one of these emission lines of a lithium atom using the shell model.
- Solve the following mapping task in which you have to assign the correct numerical values to the terms.
- Draw the principle of the formation of an emission line during flame colouring. Use the model of a lithium atom as a starting point and divide the process into meaningful sections.
If you have difficulties, look at one tip at a time.
Explanation of ionisation energies with the shell model
Task 7.3.4
We have starting from the phenomenon of different ionisation energieswe have worked outhow the shell of an atom is constructed. You should now link this knowledge.
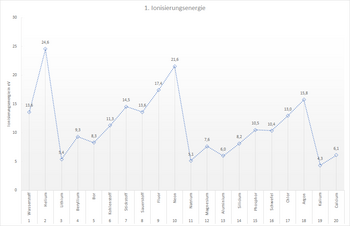
To help you solve this task, the diagram from module 7.2 is shown below. You can also draw the shell models of the respective atoms. Perhaps this will help you with the solution.
- Using the shell model of the electron shell and the examples given, explain why...
- ... the ionisation energy decreases so much with the start of a new period. Example: From neon (21.6 eV) to sodium (5.1 eV).
- ... the ionisation energy within a main group decreases from top to bottom. Example: Lithium (5.4 eV), sodium (5.1 eV) and potassium (4.3 eV).
- ... the ionisation energy increases overall from left to right within a period. Example: From lithium (5.4 eV) to neon (21.6 eV).
Task 7.3.5
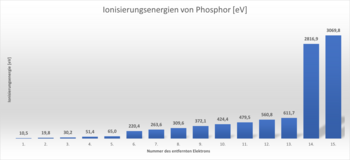
In Unit 7.2, we looked at the ionisation energies for the electrons of a phosphorus atom. You should now use your knowledge of the shell model of the atomic shell to explain this.
To help you solve this task, the diagram from Building Block 7.2 is shown below. You can also draw the shell model of a phosphorus atom. This may help you with the solution.
- Use the shell model of the electron shell to explain the course of these ionisation energies. Pay particular attention to the energy required to remove the 5th and 6th as well as the 13th and 14th electrons.
The noble gases
Octet rule
They used to be more common, but nowadays you hardly ever see them, but perhaps you still remember them: the light bulb with a filament. The picture on the right shows an illuminated light bulb.
The wire is made of tungsten. However, this reacts with the oxygen in the air, especially when it gets hot and glows. This causes the filament to break relatively quickly and the lamp no longer lights up. When the incandescent lamp was developed, attempts were made to prevent this by pumping the lamp empty of air. But even these lamps burnt out at some point because air could still penetrate from outside.
The solution was the so-called noble gases: helium, neon, argon, krypton and xenon. These elements were discovered towards the end of the 19th century. The special thing about these elements is that they only react with other substances with great difficulty, almost not at all. They are inert or "noble" and were therefore the ideal filling for incandescent lamps. The filament no longer burnt out as long as the bulb was tightly sealed.
But why are noble gases so inert?
You can explain this with the help of the shell model of the electron shell: All noble gases have a fully occupied outer electron shell. Each helium atom has 2 electrons. They occupy the first energy level or the first electron shell, which is therefore full. Neon atoms have 10 electrons, 2 on the first shell and 8 on the second shell, which is therefore also full. Atoms that have a full outer electron shell are particularly stable. This rule is called the octet rule.
If you have worked on building block 7.2, you may have already noticed something. The noble gases are also the elements with the highest ionisation energies. One reason for this is the stability of their electron shell.
For those interested in thinking ahead
Task 7.3.6
So far you have learnt about the distribution of electrons on the first 3 energy levels or in the first 3 shells of the atomic shell.
According to the distribution principle shown here, how many electrons should be in the 4th energy level or in the 4th shell of the atomic shell?
Tips
- Compare the assignment of the electrons to the energy levels with the position of the elements in the periodic table.
- For each additional element that you want to describe in the shell model, an electron is added to the model.
Resolution
According to the above diagram, there should be 18 electrons for the elements potassium to krypton (19 to 36) at energy level 4.
However, this would mean that the elements from gallium to krypton (31 to 36) would not have the same number of electrons on their outermost shell as their "upper neighbours" in periods 1 to 3. Bohr recognised a contradiction in this, as experiments on the behaviour of the elements showed that this must be the case.
He solved this problem by introducing sub-stages. The electrons of elements 21 to 30 are therefore located on their own shell, on which 10 electrons can reside. Only when this subshell is full is energy level 4 filled up further. This can be illustrated using an example:
The element arsenic has 33 electrons in its shell. The first 18 electrons are located on the first three shells, as you know from the picture, the table and the exercises. Electrons 19 and 20 are at energy level 4. The 21st electron is bound to a subshell, as are the 22nd, 23rd and so on until the subshell is filled with 10 electrons. From the 31st electron onwards, energy level 4 is filled up again until there are a total of 5 electrons on it, so that there are also 5 electrons on the top energy level of the element arsenic. This lower level introduced by Bohr could be called 4a or 4.1.
You did it, great!
You have reached the end of the unit. In module 2 you will find another section that you can now work on. You can also take the final quiz.