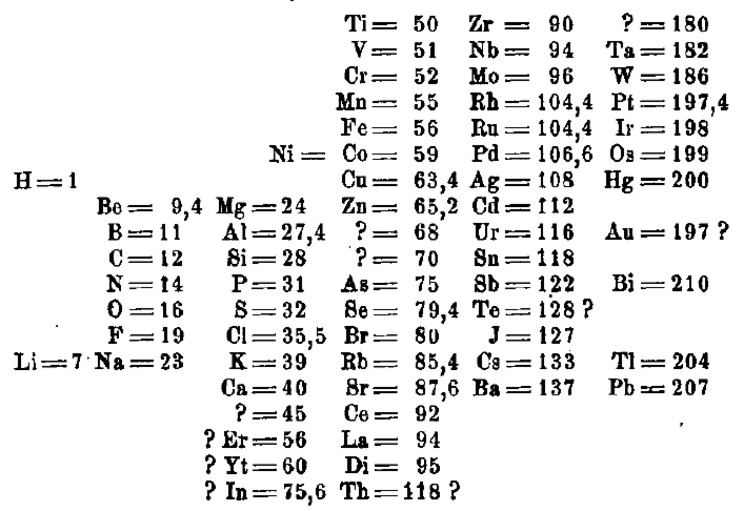Module 2: The order of the elements
You probably remember John Dalton. In 1808, he was one of the first scientists to attempt to organise the elements known at the time. He used the atomic mass known at the time as a starting point. The following table is taken from a translation of his work "A New System of Chemical Philosophy". This is how Dalton organised the elements over 200 years ago.
You have already learnt a lot about the elements and you have probably also noticed that Dalton has some "elements" in his order that do not exist today. This is because the names have changed or it has been realised that they are actually compounds.
If you compare the atomic masses of the elements with the exact values, there are also clear differences here. However, Dalton's list was the beginning of a classification system for the elements.
At the beginning of the 19th century, around 28 elements were known. Over the course of the 19th century, many more elements were discovered, their atomic masses determined and other properties characterised, so that by the end of the century their number had grown to 84. The number had therefore tripled.
The ever-growing list of elements during this period led to various scientists carrying out experiments and developing different systems for organising the elements in order to create a sensible and clear classification system.
Task 2.1
Take on the role of a scientist.
Use the 34 element cards to develop a sensible and clear classification system for the elements. To do this, identify similarities, differences and trends in the properties of the elements.
If you get stuck, the tips below may be able to help you.
Tips
1st tip
Dalton initially categorised the elements according to their atomic mass. Perhaps you could start with that too.
Task 2.2
When you are satisfied with your classification system of elements, read the section "Historical development of a classification system" and compare the classification system you have developed with the historical variants you find in this section.
Points of comparison could be:
- The atomic masses of the elements
- Elements
- Number of elements
- Are there elements that only appear in your system or only in the historical variants?
- Organisational principle
- Where can you find the elements with similar important compounds?
Historical development of an organisational system
The introductory text has already mentioned that a large number of chemical elements were discovered in the course of the 19th century. The following overview gives an idea of how quickly the number of elements increased.
| up to the year 1700 | 13 elements |
| at the end of the 18th century | 28 elements |
| by the year 1850 | 59 elements |
| at the end of the 19th century | 84 elements |
The development of a classification system for the elements seemed necessary due to the large number of elements in order to be able to use a systemic overview. The increasingly precise atomic masses and the known chemical similarities of some elements offered starting points for the creation of such an overview.
Two chemists in particular stood out in the second half of the 19th century: the German Julius Lothar Meyer and the Russian Dimitrij Iwanowitsch Mendelejew. They independently developed a classification system for the elements based on chemically similar behaviour, increasing atomic masses and periodic regularities. Mendeleev proceeded in much the same way as you have done. He reported something like this:
"Then, having written down the elements with their atomic weights and basic properties on separate cards, I began to collect the similar elements and atomic weights that were close to each other - this quickly led me to the conclusion that the properties of the elements are periodically dependent on their atomic weight." (Walden, 1965)
He published his first version of the periodic table in 1869. Subsequently, in 1870, Meyer also published his own system, which he had designed in 1868. Both scientists thus influenced each other's work, as Mendeleev then revised his system and published a new version of his periodic table of the elements in 1871.
However, Mendeleev also left gaps in his system. These gaps left room for elements yet to be discovered, whose properties (atomic mass, density, chemical behaviour, etc.) he predicted with the help of his system.
Over the next 15 years, these three elements were actually discovered: scandium, gallium and germanium. You have already categorised gallium and germanium in your system. Mendeleev's predictions about their properties were almost perfect. This was his greatest achievement with regard to the periodic properties of the elements.
The following illustrations show you the classification systems of Mendeleev and Meyer.
The noble gases
When comparing your own classification system with those of Mendeleev and Meyer, you will have realised that you have included a whole group of elements in your system that the two scientists did not integrate into their system: the noble gases (helium, neon, argon, krypton, xenon (and radon)).
These gases were discovered and recognised as elements between 1868 and 1900.
As a completely new group, they fitted perfectly into the classification system established by Mendeleev and Meyer. From this time at the latest, the periodic table was recognised as a model for ordering the elements in the natural sciences.
The periodic table today
If you have already worked up to module 7.3...
Atomic number and atomic number
The order of the elements in the periodic table is described by the atomic number. The atomic number, also known as the nuclear charge number, indicates the number of protons in an atomic nucleus. As the number of protons in the nucleus of every uncharged atom corresponds to the number of electrons in the atomic shell, the atomic number also indirectly indicates the number of electrons in an atom. For example, the element hydrogen has the atomic number 1, which means that the nucleus of the hydrogen atom consists of only one proton; there is one electron in its atomic shell. Hydrogen is followed in the periodic table by helium with atomic number 2, which means that a helium atom has two protons in its nucleus and two electrons in its atomic shell.
Task 2.3
Elements that are in the main groups of the periodic table have very similar important compounds. They are similar in their chemical behaviour.
In the shell model of the electron shell, there is always a certain number of electrons on the outermost shell of an element.
- Using the figure and the table on electron distribution, make an educated guess as to why the elements of the main groups (for example: lithium, sodium, potassium, ... or fluorine, chlorine, bromine, ...) are similar in their chemical behaviour.
- Explain why the noble gases occupy a special position in the periodic table.
Electron distribution
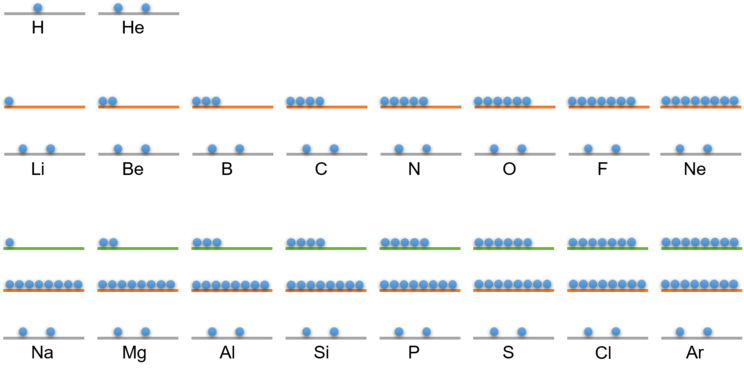
The following table shows you the distribution of electrons on the shells for the first 18 elements:
Electrons on shell 1 | Electrons on shell 2 | Electrons on shell 3 | |
1 H - hydrogen | 1 | ||
2 He - helium | 2 | ||
3 Li - Lithium | 2 | 1 | |
4 Be - Beryllium | 2 | 2 | |
5 B - boron | 2 | 3 | |
6 C - Carbon | 2 | 4 | |
7 N - Nitrogen | 2 | 5 | |
8 O - Oxygen | 2 | 6 | |
9 F - Fluorine | 2 | 7 | |
10 Ne - Neon | 2 | 8 | |
11 Na - sodium | 2 | 8 | 1 |
12 Mg - magnesium | 2 | 8 | 2 |
13 Al - aluminium | 2 | 8 | 3 |
14 Si - Silicon | 2 | 8 | 4 |
15 P - Phosphorus | 2 | 8 | 5 |
16 S - Sulphur | 2 | 8 | 6 |
17 Cl - chlorine | 2 | 8 | 7 |
18 Ar - argon | 2 | 8 | 8 |
Resolution: Electron configuration and periodic table
The structure of the electron shell is decisive for the similar chemical behaviour. In the periodic table, the elements are therefore also arranged according to the structure of their electron shell, whereby the order is determined by the atomic number.
All elements that have the same number of shells in the electron shell form a period, i.e. a row in the periodic table. There is a maximum of two electrons in the 1st shell. Therefore, there are only two elements in the 1st period: Hydrogen has one electron, helium has two electrons.
The 2nd shell can be occupied by a maximum of eight electrons. The 2nd period therefore contains eight elements from lithium (Li) to neon (Ne). The occupation of the 3rd shell begins with the element sodium (Na).
The distribution of electrons over the individual shells is called the electron configuration (electron arrangement or distribution). You worked out how to represent this in Building Block 7.2.
The sodium atom therefore has an electron in the outer shell. Similarly, the atoms of hydrogen and the other alkali metals also have an electron in the outer shell. They are in a column below each other and form the 1st main group. However, hydrogen has a special position: hydrogen is not a metal and does not behave chemically like the other elements in the 1st main group. Elements with two electrons in the outer shell form the 2nd main group (exception: helium). This principle applies to all main groups.
The noble gases occupy a special position in the periodic table as their outermost shell, the valence shell, is fully occupied with electrons. This state is called the noble gas configuration and is particularly stable. This is why noble gases form (almost) no compounds with other elements.