Module 7.2: The ionisation energy
In Building Block 3, you learnt that electrons are the building blocks of atoms. They carry a negative charge and can be moved relatively easily. Today we know that the nucleus of an atom is positively charged and that the electrons are located in the atomic shell. The nucleus and the electrons therefore attract each other because they have different charges.
Example: The hydrogen atom
Hydrogen is the simplest element. Hydrogen atoms have a positively charged proton in the nucleus, which attracts the one negatively charged electron in the atomic shell. Overall, a hydrogen atom is neutral.
This electron is located at a certain distance from the nucleus. We can compare it to a stone lying on the ground. We can lift this stone. However, energy must be supplied to do this. The more energy we supply, the higher we can lift the stone.
The same applies to electrons. We can supply them with energy and thus increase the "height", the distance from the atomic nucleus. You can lift a stone as high as you like if you supply it with enough energy. It can reach any height, any energy state.
If enough energy is utilised, electrons can even be removed from the atoms. Today, scientists can measure the energy required to remove an electron from an atom very precisely.
The energy required to detach an electron from the attractive force of the positively charged nucleus is called ionisation energy.
Ionisation energies of the elements
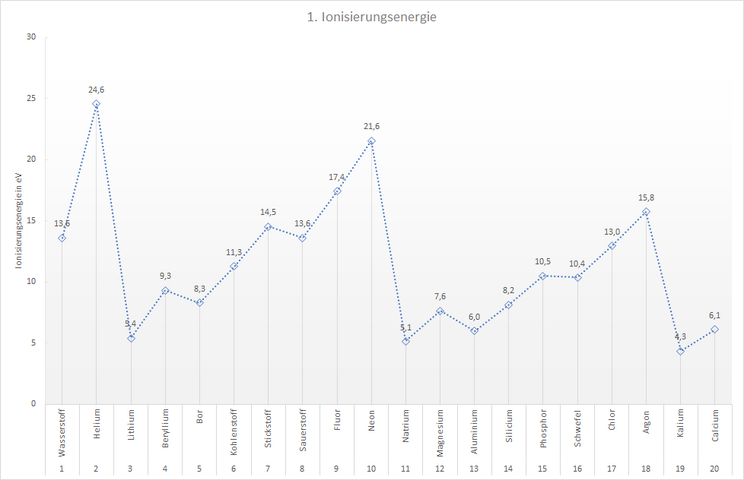
The following table lists the minimum amount of energy required to remove exactly one electron from an atom of an element. This 1st ionisation energy is shown for the first 20 elements.
Note: The unit electron volt [eV] is the "handy" unit of energy when very small amounts of energy need to be specified. 1 eV is 1.602 - 10-19 J - s. That is very little energy. If you want to heat 1 g of water by 1 degree, you would need the energy 2.609 - 1019 eV.
| Element | Ionisation energy in eV |
|---|---|
| Hydrogen | 13,6 |
| Helium | 24,6 |
| Lithium | 5,4 |
| Beryllium | 9,3 |
| boron | 8,3 |
| Carbon | 11,3 |
| Nitrogen | 14,5 |
| Oxygen | 13,6 |
| Fluorine | 17,4 |
| Neon | 21,6 |
| Sodium | 5,1 |
| magnesium | 7,6 |
| Aluminium | 6,0 |
| Silicon | 8,2 |
| Phosphorus | 10,5 |
| Sulphur | 10,4 |
| Chlorine | 13,0 |
| Argon | 15,8 |
| Potassium | 4,3 |
| Calcium | 6,1 |
| ... |
Task 7.2.1 - for professionals (optional)
Draw a diagram by hand or using Excel, for example, which shows the progression of the ionisation energy of the first 20 elements.
Ionisation energy curve in the periodic table
Task 7.2.2
- For professionals (optional): Formulate a written description of the diagram.
- Tip: The worksheet "Strategy for describing a diagram" can help you with this.
- Make sure that you only describe and do not analyse.
- If you are not doing the task for professionals, solve the digital task (see below). If you fill in the gaps in the text with the correct words, you will receive a description of the diagram
- Use the diagram to determine rules that describe the rough progression of the ionisation energy within the periodic table (if you need help, use the tip below):
- From top to bottom (within a main group)...
- From left to right (within a period)...
Tip for subtask 3
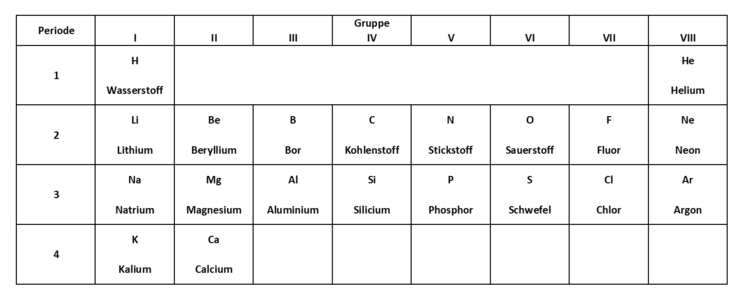
This abridged version of the periodic table can help you to solve subtask 3.
Take a look at the diagram or the table of ionisation energies. You can pick out the ionisation energy for an electron from the table to match the elements. Using the shortened version of the periodic table, you can then solve the task more easily.
Marking: The ionisation energy is the amount of energy required to remove an electron of the corresponding element from the atomic shell.
For interested parties
Special conditions
You have probably already noticed that the ionisation energy does not increase evenly from left to right within a period, but that there are elements for which the ionisation energy has a clear "kink". They have a lower ionisation energy than the element before them. They do not fit into the trend.
In the 2nd period these are the elements boron and oxygen, in the 3rd period aluminium and sulphur. This trend continues in the following periods: The elements of the 3rd main group and those of the 6th main group have significantly lower ionisation energies than would be assumed. The 1st electron can therefore be removed more easily in these elements, it is less strongly bound to the atom than in the "neighbouring elements".
The diagram shows us that the electrons that can first be removed from the atom of an element must be bound to the nucleus to different degrees. To remove the first electron from a neon atom, we need almost twice as much energy (21.6 eV) as for the first electron from a carbon atom (11.3 eV). The atomic shells of the elements must therefore differ from each other in some way.
In order to investigate the structure of the atomic shell, we will now take a closer look at the ionisation energies of an element. So far we have only looked at the energy required to remove an electron from the atom. If one electron is to be removed from the atom at a time, the energy for each electron can be determined until all electrons have been removed from the atom.
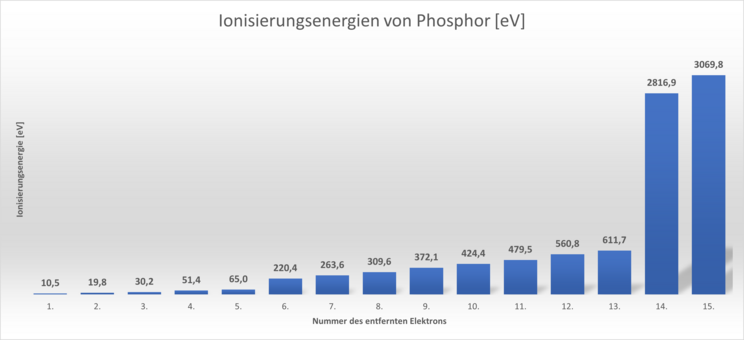
The following diagram shows these ionisation energies for phosphorus. A phosphorus atom has 15 electrons.
Task 7.2.3
- Formulate a written description of the diagram.
- Tip: The worksheet "Strategy for describing a diagram" can help you with this.
- Make sure that you only describe and do not analyse.
The bars in the diagram always correspond to the amount of energy required to split off the 1st, 2nd, 3rd etc. electron. electron.
We can see that the splitting off of each additional electron becomes increasingly difficult. This is also easy to explain: Fewer and fewer electrons are attracted to the positive charge of the nucleus. This remains the same. It is striking that the increase makes a large jump after the 5th and 13th electron.
This shows us that the electrons in an atom differ from each other in their energy content. They are said to be at different energy levels. Apparently, therefore, there are energy levels from which the electrons are easier to detach than from other energy levels.
To visualise these energy levels, imagine that an electron is a stone on a ladder with a certain number of steps. If a stone is on a low step (i.e. it is close to the ground), a lot of energy is still needed to lift the stone up all the steps of the ladder. If you expend enough energy, at some point the stone is no longer on the ladder, it has been completely removed. In terms of an atom, this means that if the electron is at a low energy level, i.e. close to the nucleus, a lot of energy is required to remove it from the atomic shell. If enough energy is applied, the electron can be removed completely, just like the stone, and is then no longer part of the atomic shell.
But now the question arises: How can we visualise these energy levels in more detail and how are the electrons actually distributed if there is more than one electron in the atomic shell?
You will find the answer to this question in Building Block 7.3.