-
 Table: Nuclear charge and atomic mass This table contains the elements with atomic charge 1 to 84 and their atomic mass.
Table: Nuclear charge and atomic mass This table contains the elements with atomic charge 1 to 84 and their atomic mass. -

In-depth station: Nuclear charge and atomic mass
In the two stations of this module, you have learnt more about the nucleus of an atom and its structure. Atomic nuclei are made up of protons and neutrons. The number of protons indicates the atomic number because protons are simply positively charged. A neutron is not charged, but has the same mass as a proton. You have also learnt that the mass of an atom is determined almost exclusively by the number of protons and neutrons. The mass of the electrons only makes up a very small proportion of the total mass of an atom. In the model experiment, you were also able to feel for yourself what role the neutrons play in the atomic nucleus: they ensure that the nucleus holds together and does not disintegrate due to the repulsive forces between the protons.
We can now ask ourselves: How is the number of protons, i.e. the nuclear charge number, related to the number of neutrons in the nucleus? How many neutrons does a nucleus "need" to remain stable?
To analyse this relationship, you can use the nuclear charge number and the atomic mass. The atomic number is the number of protons in the nucleus and the atomic mass is the number of protons + the number of neutrons.
Task 6.3.1 - for professionals (optional)
Draw a diagram by hand or using Excel, for example, that shows the relationship between nuclear charge and atomic mass. Use different elements, at least 25 of them. You can find the data for this under Material on the left-hand side. Also draw the straight line that would result if the atomic mass of each element were twice as large as its atomic number.
The following table can serve as a guide for you:
| Element | Nuclear charge | Atomic mass (u) |
|---|---|---|
| Hydrogen | 1 | 1,0079 |
| Helium | 2 | 4,0026 |
| Lithium | 3 | 6,9675 |
| Beryllium | 4 | 9,0122 |
| boron | 5 | 10,813 |
| Carbon | 6 | 12,011 |
| ... |
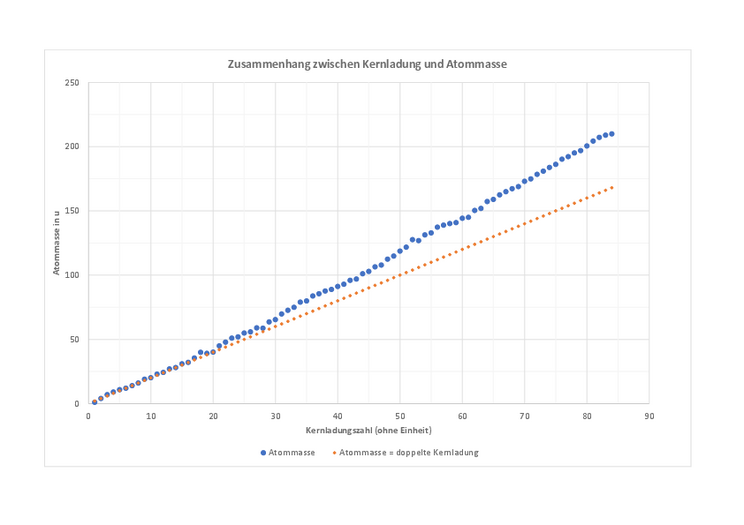
The relationship between nuclear charge and atomic mass
This diagram shows the relationship between the atomic charge and the atomic mass of all elements up to atomic number 84. It also shows the straight line that would result if the atomic mass of each element were twice its atomic number. If you have done the optional task, your diagram could look something like this, only with fewer points.
Task 6.3.2
- Formulate a written description of the diagram.
- Tip: The worksheet "Strategy for describing a diagram" can help you with this.
- Make sure that you only describe and do not analyse.
- When you have finished your description, complete the digital task. If you solve this correctly, you will have a sample solution for the diagram description. Use it to check your own description and correct it if necessary.
Task 6.3.3: Analysing the diagram
Now that you have described the diagram, you need to analyse it. At the beginning of this station, we asked ourselves:
How is the number of protons, i.e. the nuclear charge number, related to the number of neutrons in the nucleus? How many neutrons does a nucleus "need" to remain stable?
Interpret the relationship between nuclear charge and atomic mass shown in the diagram in relation to these questions. Use your diagram description and the summary from the beginning of this station. First formulate your interpretation, then you can look at the sample solution and compare it with your interpretation.
Proposed solution Diagram evaluation
Protons and neutrons have almost exactly the same mass, but only the protons determine the nuclear charge. Atomic mass minus nuclear charge number gives approximately the neutron number.
This means that the orange data series shows how large the atomic mass of the elements would be if the number of neutrons were always the same as the number of protons. The diagram shows that this relationship only applies to light elements with a low proton number. The higher the atomic number, i.e. the number of protons in the nucleus, the more the atomic mass deviates from the orange data series. This means that more neutrons than protons are "necessary" for the atomic nucleus of heavy elements to remain stable.