Contact
Dr. Bert Engelen

Institut für Chemie und Biologie des Meeres (ICBM) (» Postanschrift)
Prof. Dr. Heribert Cypionka
Deep sea drilling projects (ODP/IODP) (2002-2016)
Impact of salinity changes on viral production during the paleoenvironmental history of Baltic Sea sediments
The Baltic Sea is an intra-continental basin that has undergone alterations between limnic, brackish and marine conditions due to repeated glaciation and sea-level changes in the past. The sediments represent an archive of this paleoenvironmental history. Recently, sediments from the Baltic Sea were recovered during IODP Exp. 347 with the aim to identify the impact of the dramatic variations in salinity on the microbial communities within the deep subsurface.
In the this project, we will focus on viruses and their host organisms as relevant components of the deep biosphere. As viruses control microbial populations and mediate enhanced carbon turnover due to lysis of infected host cells, they have a major impact on microbial and biogeochemical processes.
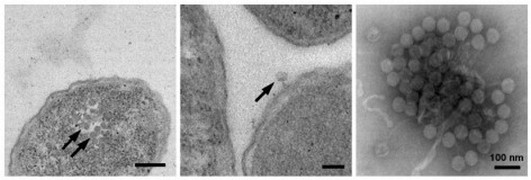
In our previous investigations, viruses have been detected in sediments as old as 14 Ma and as deep as 320 mbsf. Increasing virus-to-cell ratios with depth indicated ongoing viral production in the deep subsurface. However, as only little is known about general characteristics of benthic viruses, in this project, we will investigate viral production, decay, life modes and induction mechanisms in Baltic Sea sediments.
We will directly measure rates of viral production and further determine if salinity changes triggered the induction of prophages in the past. By analyzing different sediment layers, enrichment cultures and isolates, we will determine if this variation has influenced prokaryotic and viral communities' structures. With our study, we will gain deeper insights into the impact of viruses on current subsurface communities and their role during the paleoenvironmental history of the Baltic Sea.
Team
- Engelen B, Engelhardt T, Cypionka H (2014) Phagen in Sedimenten der marinen tiefen Biosphäre. Biospektrum 04.14:380-382, DOI: 10.1007/s12268-014-0451-0
- Engelhardt T, Kallmeyer J, Cypionka H, Engelen B (2014) High virus-to-cell ratios indicate on-going production of viruses in deep subsurface sediments. ISME J 8:1503–1509
- Engelhardt T, Sahlberg M, Cypionka H, Engelen B (2013) Biogeography of Rhizobium radiobacter and distribution of associated temperate phages in deep subseafloor sediments. ISME J 7:199-209
- Engelhardt T, Sahlberg M, Cypionka H, Engelen B (2011) Induction of prophages from deep-subseafloor bacteria Environm Microbiol Rep 3:459–465
Related Publications
- Engelen B, Engelhardt T, Cypionka H (2014) Phagen in Sedimenten der marinen tiefen Biosphäre. Biospektrum 04.14:380-382, DOI: 10.1007/s12268-014-0451-0
- Engelhardt T, Kallmeyer J, Cypionka H, Engelen B (2014) High virus-to-cell ratios indicate on-going production of viruses in deep subsurface sediments. ISME J 8:1503–1509
- Engelhardt T, Sahlberg M, Cypionka H, Engelen B (2013) Biogeography of Rhizobium radiobacter and distribution of associated temperate phages in deep subseafloor sediments. ISME J 7:199-209
- Engelhardt T, Sahlberg M, Cypionka H, Engelen B (2011) Induction of prophages from deep-subseafloor bacteria Environm Microbiol Rep 3:459–465
Viral impact on microbiology and geochemistry of extremly nutrient-depleted sediments
The overall goal of our project is to understand the impact of phages for biogeochemical cycling, microbial abundance and diversity in the marine subsurface. The integration of the viral component into the biogeochemical model for subsurface sediments of the South Pacific Gyre (SPG) is essential to understand the recycling of labile organic matter within this habitat. Thus, our aim is to identify if viral lysis could provide these compounds to this extremely nutrient-depleted habitat. We hypothesise that viruses might be one controling factor for the abundance and diversity of indigenous microorganisms.
First, viral counts were determined on SPG sediments to calculate their distribution within the subsurface. Our results indicate that the number of viruses always exceeded those of prokaryotic cells with an increasing virus-to-cell ratio with depth.
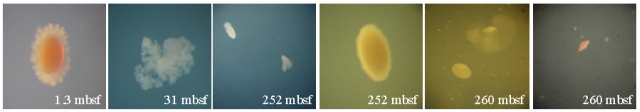
In our cultivation-based approach, we have started to isolate subseafloor microorganisms from the SPG to identify prophages within their genomes. Sediment samples that were recovered from various sites and sediment horizons were directly transferred into three different media onboard the JOIDES Resolution. Two media containing a mix of several monomers as substrates were used to target either aerobic or anaerobic heterotrophs. For the enrichment of autotrophs, cultures were set up in artifical seawater medium (without any organic substrate) and flushed with hydrogen/carbon dioxide.
Due to the slow growth of indigenous microorganisms, we have now obtained the first isolates from a limited number of oxic enrichments from site U1371 (3.7 mbsf, 122 mbsf, 127 mbsf). Those are affiliated to Nocardioides basaltis (99% 16S rRNA sim.), Halomonas aquamarina (100%), Erythrobacter vulgaris (99%), Pseudoaltoromonas sp. (99%), Alteromonas sp. (100%), and Dietzia sp. (100%). Their presence was already detected by molecular screening of the cultures by DGGE. When the process of strain purification is completed, we will start to induce the prophages within their genomes. The morphologic and phylogenetic diversity of these phages will be analysed in detail. Concerning our culture collection, we will then identify if the percentage of infected cells that are under severe starvation is different to those isolated from "high activity sites".
The diversity of viruses within the SPG sediments will be analysed by applying a novel approach using whole genome amplification. This technique is required to enhance the amount of viral DNA in sediments with an extremly low abundance of viruses. Subsequently, other molecular down stream applications such as RAPD-PCR will be used to analyse virus diversity patterns of the different sites.
Team
- Franziska Preuss (PhD student)
- Lisa Marie Moskwa (Master student)
- Bert Engelen
- Heribert Cypionka
Related publications
- Engelen B, Engelhardt T, Cypionka H (2014) Phagen in Sedimenten der marinen tiefen Biosphäre. Biospektrum 04.14:380-382, DOI: 10.1007/s12268-014-0451-0
- Engelhardt T, Kallmeyer J, Cypionka H, Engelen B (2014) High virus-to-cell ratios indicate on-going production of viruses in deep subsurface sediments. ISME J 8:1503–1509
- Engelhardt T, Sahlberg M, Cypionka H, Engelen B (2013) Biogeography of Rhizobium radiobacter and distribution of associated temperate phages in deep subseafloor sediments. ISME J 7:199-209
- Engelhardt T, Sahlberg M, Cypionka H, Engelen B (2011) Induction of prophages from deep-subseafloor bacteria Environm Microbiol Rep 3:459–465
Viral infections as controlling factors of the deep biosphere
The marine deep biosphere represents the largest biotope on Earth. Throughout the last years, we have obtained interesting insights into its microbial community composition. However, one component that was completely overlooked so far is the viral inventory of deep-subsurface sediments. As grazers probably play only a minor role in anoxic and highly compressed deep sediments, viruses might be the main "predators" for indigenous microorganisms. Furthermore, the viral shunt could have a major impact on the deep biosphere in providing labile organic compounds to the microbial community in this generally nutrient depleted habitat.
From cultivation experiments on samples recovered during ODP Leg 201 and IODP Exp. 301 we have obtained large culture collections of deep-biosphere isolates. These culture collections represent an archive of the viral composition within the deep biosphere as up to 70% of cultivated bacteria contain prophages within their genome. In six out of thirteen representative isolates, prophages were induced to form free virus-particles that will be analysed in detail due to their morphologic and phylogenetic diversity.
In this project, we will identify the phage inventory of the deep biosphere by ecophysiological studies on ODP/IODP isolates to answer the following questions: To which extent are deep-biosphere populations controlled by viral infections? What is the inter- and intraspecific diversity and the host-specific viral biogeography? Can viral infections tell us something about the physiological state of indigenous microorganisms? Finally, we will obtain estimates for the viral shunt as an important factor for sustaining the deep biosphere.
Team
- Tim Engelhardt
- Monika Sahlberg
- Wiebke Landwehr
- Bert Engelen
- Heribert Cypionka
Related publications
- Engelen B, Engelhardt T, Cypionka H (2014) Phagen in Sedimenten der marinen tiefen Biosphäre. Biospektrum 04.14:380-382, DOI: 10.1007/s12268-014-0451-0
- Engelhardt T, Kallmeyer J, Cypionka H, Engelen B (2014) High virus-to-cell ratios indicate on-going production of viruses in deep subsurface sediments. ISME J 8:1503–1509
- Engelhardt T, Sahlberg M, Cypionka H, Engelen B (2013) Biogeography of Rhizobium radiobacter and distribution of associated temperate phages in deep subseafloor sediments. ISME J 7:199-209
- Engelhardt T, Sahlberg M, Cypionka H, Engelen B (2011) Induction of prophages from deep-subseafloor bacteria Environm Microbiol Rep 3:459–465
Influence of fluids from the ocean crust on growth and activity of deep-biosphere populations (IODP Leg 301)
The goal of our investigations is to understand the role of crustal fluids as a driving force for the marine deep-biosphere. To analyze microbial activities and community structures, contamination-free sediment samples were recovered during IODP Leg 301 to the Juan de Fuca Ridge off the coast of Oregon.
The sediment of IODP Site 1301 (water depth: 2665 m, sediment thickness: 265 m) is characterized by a diffusive flow of fluids from the underlying oceanic crust, a steep temperature gradient of 0.23 °C/m, and two sulfate-methane transition zones.
The hypothesis that this geochemical regime of an "upside down" electron acceptor series fuels the deep biosphere was proven by activity measurements and the quantification of cells along the sediment column. Total cell counts and the number of 16S rRNA genes increased within the sulfate-methane transition zones and above the basement. Potential phosphatase activity was enhanced in phosphate-depleted layers towards the sediment-basement interface. Rates of sulfate reduction and anaerobic oxidation of methane (AOM) were elevated within the lower sulfate-methane transition zone.
Further investigations focused on isolating indigenous microorganisms. Microbial growth was stimulated in various enrichment cultures from the entire sediment column. Molecular screening via PCR-DGGE, used to determine the microbial composition of enrichments, revealed a broad set of different phylotypes. These data allow a guided isolation of unique deep-biosphere microorganisms. A subsequent characterization will determine special adaptations to their habitat.
Team
- Katja Ziegelmüller
- Falko Mathes
- Lars Wolf
- Bert Engelen
- Heribert Cypionka
Related publications
- Fichtel K, Mathes F, Könneke M, Cypionka H, Engelen B (2012) Isolation of sulfate-reducing bacteria from sediments above the deep-subseafloor aquifer. Front Microbio 3:65
- Engelen B and Cypionka H (2008) The subsurface of tidal flats as a model for the deep biosphere. Ocean Dynamics 59:385–391
- Engelen B, Ziegelmüller K, Wolf L, Köpke B, Gittel A, Treude T, Nakagawa S, Inagaki F, Lever MA, Steinsbu BO, Cypionka H (2008) Fluids from the oceanic crust support microbial activities within the deep biosphere. Geomicrobiology J 25:56-66
- Ziegelmüller K, Könneke M, Cypionka H, and Engelen B (2007) Cultivation of sulfate-reducing bacteria from deep sediment layers that are influenced by crustal fluids (IODP Leg 301) Extended abstract, IODP/ICDP-Kolloquium Potsdam
- Ziegelmüller K, Cypionka H, and Engelen B (2007) Hydrothermal fluids from the oceanic crust stimulate metabolic activities of deep-biosphere populations (IODP Leg 301). Extended abstract, IODP/ICDP-Kolloquium Potsdam
- Lever MA, Alperin M, Engelen B, Inagaki F, Nakagawa S, Steinsbu BO, Teske A, IODP Expedition 301 Scientists (2006) Trends in basalt and sediment core contamination during IODP expedition 301. Geomicrobiol J 23:517-530
- Nakagawa S, Inagaki F, Suzuki Y, Steinsbu BO, Lever MA, Takai K, Engelen B, Sako Y, Wheat CG, Horikoshi K, and IODP Exp. 301 Scientists (2006) Microbial Community in Black Rust Exposed to Hot Ridge Flank Crustal Fluids. Appl Environ Microbiol 72:6789–6799
Links
- Official IODP Leg 301 webpage
- Reisebericht von Bert Engelen Cruise report (in German)
- Teacher at Sea (Detailed cruise report)
Deep-biosphere bacteria from ODP Leg 201
Marine sediments harbour a considerable part of the living biomass on earth. The microbial communities within these sediments are characterised by low activities and extremely slow growth. Anyhow, increased cell numbers and activities in upper sulfate-methane transition zones (21-40 meter below seafloor) and in lower sulfate-methane transition zones (90 mbsf) demonstrate the presence of active microbial communities within pacific sediments. To get insights how these microbes thrive within their habitat, isolation and characterisation of dominant representives is necessary. Therefore, novel cultivation approaches were applied to contamination-checked sediment samples taken from the Equatorial Pacific and Peru Margin during ODP Leg 201 of JOIDES Resolution in Jan. – Apr. 2002.
Team
- Anja Batzke
- Henrik Sass
- Bert Engelen
- Heribert Cypionka
Related publications
- Engelen B and Cypionka H (2008) The subsurface of tidal flats as a model for the deep biosphere. Ocean Dynamics DOI 10.1007/s10236-008-0166-1
- Batzke A, Engelen B, Sass H, Cypionka H (2007) Phylogenetic and physiological diversity of cultured deep-biosphere bacteria from Equatorial Pacific Ocean and Peru Margin sediments. Geomicrobiology J 24:261-273
- Cypionka H, Engelen B, Sass H, ODP Leg 201 Scientific Shipboard Party (2002) Novel approaches to the cultivation of marine sediment bacteria. Geochim Cosmochim Acta 66:A161
- Cypionka R, Cypionka H (2004) Leben in der Erdkruste - eine Forschungsreise in die "tiefe Biosphäre". Unterricht Biologie 299:26-28
- Cypionka R, Cypionka H (2004) Reise in die tiefe Biosphäre. Unterricht Biologie 299:29-32
- D'Hondt S, Jørgensen BB, Miller DJ, Batzke A, Blake R, Cragg BA, Cypionka H, Dickens GR, Ferdelman T, Hinrichs KU, Holm NG, Mitterer R, Spivack A, Wang G, Bekins B, Engelen B, Ford K, Gettemy G, Rutherford SD, Sass H, Skilbeck CG, Aiello IW, Guèrin G, House C, Inagaki F, Meister P, Naehr T, Niitsuma S, Parkes RJ, Schippers A, Smith DC, Teske A, Wiegel J, Padilla CN, Acosta JLS (2004) Distributions of microbial activities in deep subseafloor sediments. Science 306:2216-2221
- D'Hondt SL, Jørgensen BB, Miller DJ, Aiello IW, Bekins B, Blake R, Cragg BA, Cypionka H, Dickens GR, Ferdelman T, Ford KH, Gettemy GL, Guèrin G, Hinrichs K-U, Holm N, House CH, Inagaki F, Meister P, Mitterer RM, Naehr TH, Niitsuma S, Parkes RJ, Schippers A, Skilbeck CG, Smith DC, Spivack AJ, Teske A, Wiegel J (2003) Controls on microbial communities in deeply buried sediments, eastern Equatorial Pacific and Peru margin, Sites 1225 - 1231. Proc. ODP, Init. Repts., 201 [Online]. Available from World Wide Web: www-odp.tamu.edu/publications/201_IR/201ir.htm, ISSN 1096-2158
- Sass H, Engelen B, Cypionka H (2003) Die tiefe Biosphäre - Mikrobiologie der Erdkruste. BioSpektrum 9:589-591
Links
- Microbiological garden