Kontakt
What it measures
What it measures
Traditionally, brain electrical activity is recorded from upper head scalp sites, whereas locations behind the ear are ignored, or used as reference sites only. One goal of the cluster of excellence Hearing4All is to develop brain-computer interface technology for hearing aid control. Obviously, this ambitious goal requires unobtrusive, motion-tolerant, comfortable and discreet EEG acquisition in the first place. We first evaluated information that can be obtained from miniature electrodes placed in the outer-ear canal and the concha (Bleichner et al., 2015). From this study we learned that the distance between electrodes is important; a larger inter-electrode distance allows us to capture signals from further away. Furthermore the ability to derive bipolar channels of several different orientations (see also Bleichner et al., 2016), in order to "sense" into different directions, was found to be a necessary feature. This reasoning naturally leads to a C-shaped multi-sensor array that is placed around the ear. To implement this we teamed up with the company TMSI, who have been tremendously helpful in turning our idea into a real product.
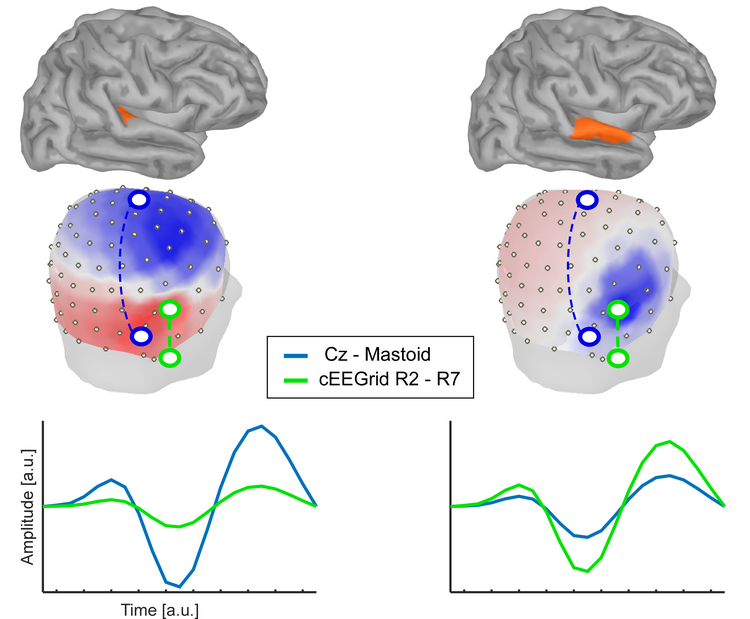
In a toy example illustrated here, we placed a single source into one of two locations, either the Heschl gyrus (left) or the superior temporal gyrus (right). The figure shows the voltage topography that would be measured with high-density EEG if only those sources were active. For illustration purposes we compare two bipolar channels, one derived from the cap-EEG data (blue) and one derived from cEEGrid EEG data (green). As can be seen in one condition the signal measured is larger for the cEEGrid channel, whereas in the other condition it is larger for the cap-EEG channel. Whether a brain source is "seen" from an EEG channel or not depends (among a couple of other reasons happily ignored here) to a large extent on whether the orientation of the EEG channel is aligned to the orientation of the dipolar source activity. In fact, a bipolar channel placed directly on top of a dipolar tangential source and oriented orthogonally to it may not capture any of the sources activity. Distance between electrodes, however, matters. Bipolar channels derived from two electrodes located very close to each other cannot "sense" very far, since the (far field) common voltage signals will be subtracted out.
Resting EEG
In Debener et al., (2015) we show that the famous Berger effect, a difference in 8-13 Hz (alpha) activity between eyes open and eyes closed resting conditions, can be easily confirmed with cEEGrid recordings. A good part of the resting EEG alpha activity originates in from occipital cortex, and is one feature captured by the cEEGrids.
Late Auditory Evoked Potentials (AEP)
Also in Debener et al., (2015) we report auditory oddball event related potentials (ERPs). In this study, stimulus presentation and EEG data collection was implemented on a single off-the-shelf smartphone. We confirmed AEP N100 deflections using this cEEGrid recording environment, despite the suboptimal stimulus timing encountered on Android operating systems. Attentional gain effects on the AEP N100 were also confirmed by Bleichner et al., (2016), even in a single-trial analysis.
P300 ERPs
In the Debener et al., (2015) smartphone study clear P300 ERP component effects were found in response to target sounds. A classification analysis revealed that it could be decoded from single-trial cEEGrid EEG whether a target or a non-target sound was presented. Note that classification accuracy in this paradigm is not good enough for supporting a BCI application, but cEEGrid classification accuracy was very similar to the multi-channel cap EEG results of another group reporting classification accuracies of oddball data.
Decoding the attended speaker with speech envelope tracking
Mirkovic et al., (2015) replicated previous work showing the attended speaker in a two-stream listening scenario could be decoded with an envelope tracking procedure. In a follow-up paper, Mirkovic et al., (2016) found that attended speaker decoding is also possible with cEEGrid EEG, albeit with some loss in performance when compared to cap EEG.
Sleep EEG
We have completed several sleep recordings and found that typical sleep EEG features (K complex, slow wave sleep, spindles, theta, etc.) are represented in cEEGrid EEG recordings. A proper validation study is under way.
Epilepsy EEG
In a very first pilot pediatric EEG study we confirmed the presence of interictal activity in cEEGrid EEG recordings, very similar to the concurrently recorded cap-EEG signals. Since key features of the cEEGrid technology are stable impedances for many hours and high wearing comfort, we believe the technology has great potential for improving current practice in pediatric epilepsy EEG acquisition. Validation studies are needed to identify the sensitivity and specificity of cEEGrid EEG signals for picking-up clinically relevant information.
Eye movements
cEEGrid signals capture eye blinks and eye movements. Note however that different referential signals are required. In order to capture eye blinks a bipolar channel of vertical orientation (e.g. R2 vs. R7) should be used. If the goal is to capture lateral eye movements a horizontal bipolar channel is needed (e.g., R1 vs. R4). See Debener et al., 2015 for details on labelling cEEgrid channels.
Electrocardiogram
In many individuals, cEEGrid EEG data are contaminated by cardiac electrical activity (EKG). We use independent component analysis to disentangle EKG from brain electrical activity (EEG). This approach can be used to attenuate artifacts in multi-channel EEG recordings and also to improve the signal to noise ratio of the EKG. The ICA-derived cEEGrid EKG thus enables the analysis of heart-rate variability measures.